Antimicrobial and antibiofilm activities of Cu(II) Schiff base complexes against methicillin-susceptible and resistant Staphylococcus aureus
- PMID: 34560892
- PMCID: PMC8464119
- DOI: 10.1186/s12941-021-00473-4
Antimicrobial and antibiofilm activities of Cu(II) Schiff base complexes against methicillin-susceptible and resistant Staphylococcus aureus
Abstract
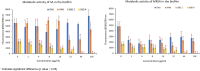
Background: Methicillin-resistance S. aureus (MRSA) possesses the ability to resist multiple antibiotics and form biofilm. Currently, vancomycin remains the last drug of choice for treatment of MRSA infection. The emergence of vancomycin-resistant S. aureus (VRSA) has necessitated the development of new therapeutic agents against MRSA. In this study, the antimicrobial and antibiofilm activities of two copper-complexes derived from Schiff base (SBDs) were tested individually, and in combination with oxacillin (OXA) and vancomycin (VAN) against reference strains methicillin-susceptible and methicillin-resistant Staphylococcus aureus. The toxicity of the SBDs was also evaluated on a non-cancerous mammalian cell line.
Methods: The antimicrobial activity was tested against the planktonic S. aureus cells using the microdilution broth assay, while the antibiofilm activity were evaluated using the crystal violet and resazurin assays. The cytotoxicity of the SBDs was assessed on MRC5 (normal lung tissue), using the MTT assay.
Results: The individual SBDs showed significant reduction of biomass and metabolic activity in both S. aureus strains. Combinations of the SBDs with OXA and VAN were mainly additive against the planktonic cells and cells in the biofilm. Both the compounds showed moderate toxicity against the MRC5 cell line. The selectivity index suggested that the compounds were more cytotoxic to S. aureus than the normal cells.
Conclusion: Both the SBD compounds demonstrated promising antimicrobial and antibiofilm activities and have the potential to be further developed as an antimicrobial agent against infections caused by MRSA.
Keywords: Biofilms; Methicillin-resistant Staphylococcus aureus; Schiff base derivatives.
© 2021. The Author(s).
Conflict of interest statement
The authors declare that they have no competing interests.
Figures
References
-
- Low ML, Paulus G, Dorlet P, Guillot R, Rosli R, Delsuc N, Crouse KA, Policar C. Synthesis, characterization and biological activity of Cu(II), Zn(II) and Re(I) complexes derived from S-benzyldithiocarbazate and 3-acetylcoumarin. Biometals. 2015;28:553–566. doi: 10.1007/s10534-015-9831-2. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases