Cholesteryl ester transfer protein (CETP) as a drug target for cardiovascular disease
- PMID: 34561430
- PMCID: PMC8463530
- DOI: 10.1038/s41467-021-25703-3
Cholesteryl ester transfer protein (CETP) as a drug target for cardiovascular disease
Abstract
Development of cholesteryl ester transfer protein (CETP) inhibitors for coronary heart disease (CHD) has yet to deliver licensed medicines. To distinguish compound from drug target failure, we compared evidence from clinical trials and drug target Mendelian randomization of CETP protein concentration, comparing this to Mendelian randomization of proprotein convertase subtilisin/kexin type 9 (PCSK9). We show that previous failures of CETP inhibitors are likely compound related, as illustrated by significant degrees of between-compound heterogeneity in effects on lipids, blood pressure, and clinical outcomes observed in trials. On-target CETP inhibition, assessed through Mendelian randomization, is expected to reduce the risk of CHD, heart failure, diabetes, and chronic kidney disease, while increasing the risk of age-related macular degeneration. In contrast, lower PCSK9 concentration is anticipated to decrease the risk of CHD, heart failure, atrial fibrillation, chronic kidney disease, multiple sclerosis, and stroke, while potentially increasing the risk of Alzheimer's disease and asthma. Due to distinct effects on lipoprotein metabolite profiles, joint inhibition of CETP and PCSK9 may provide added benefit. In conclusion, we provide genetic evidence that CETP is an effective target for CHD prevention but with a potential on-target adverse effect on age-related macular degeneration.
© 2021. The Author(s).
Conflict of interest statement
A.F.S. has received Servier funding for unrelated work. M.Z. conducted this research as an employee of BenevolentAI. Since completing the work M.Z. is now a full-time employee of GlaxoSmithKline. Since completing the work C.J.O. is now a full-time employee of Novartis Institutes for Biomedical Research. D.A.L. has received support from Roche Diagnostics and Medtronic Ltd. for research unrelated to this paper. T.R.G. receives funding from GlaxoSmithKline and Biogen.
Figures

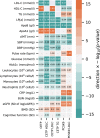


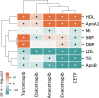

References
Publication types
MeSH terms
Substances
Grants and funding
- MR/V033867/1/MRC_/Medical Research Council/United Kingdom
- MC_UU_12019/1/MRC_/Medical Research Council/United Kingdom
- RG/10/12/28456/BHF_/British Heart Foundation/United Kingdom
- R21 HL140385/HL/NHLBI NIH HHS/United States
- 221854/Z/20/Z/WT_/Wellcome Trust/United Kingdom
- FS/17/70/33482/BHF_/British Heart Foundation/United Kingdom
- MC_UU_00011/4/MRC_/Medical Research Council/United Kingdom
- R01 HL105756/HL/NHLBI NIH HHS/United States
- RF1 AG062553/AG/NIA NIH HHS/United States
- R01 LM010098/LM/NLM NIH HHS/United States
- MC_UU_00011/6/MRC_/Medical Research Council/United Kingdom
- CH/F/20/90003/BHF_/British Heart Foundation/United Kingdom
- 29019/CRUK_/Cancer Research UK/United Kingdom
- SP/13/6/30554/BHF_/British Heart Foundation/United Kingdom
- PG/18/50/33837/BHF_/British Heart Foundation/United Kingdom
- CS/13/1/30327/BHF_/British Heart Foundation/United Kingdom
- R01 AG025533/AG/NIA NIH HHS/United States
- R01 DK117445/DK/NIDDK NIH HHS/United States
- MR/R024227/1/MRC_/Medical Research Council/United Kingdom
- MR/S011676/1/MRC_/Medical Research Council/United Kingdom
- R01 MD012765/MD/NIMHD NIH HHS/United States
- WT_/Wellcome Trust/United Kingdom
LinkOut - more resources
Full Text Sources
Miscellaneous

