The Immune Privilege of Cancer Stem Cells: A Key to Understanding Tumor Immune Escape and Therapy Failure
- PMID: 34572009
- PMCID: PMC8469208
- DOI: 10.3390/cells10092361
The Immune Privilege of Cancer Stem Cells: A Key to Understanding Tumor Immune Escape and Therapy Failure
Abstract
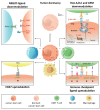
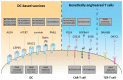
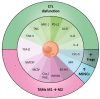
Cancer stem cells (CSCs) are broadly considered immature, multipotent, tumorigenic cells within the tumor mass, endowed with the ability to self-renew and escape immune control. All these features contribute to place CSCs at the pinnacle of tumor aggressiveness and (immune) therapy resistance. The immune privileged status of CSCs is induced and preserved by various mechanisms that directly affect them (e.g., the downregulation of the major histocompatibility complex class I) and indirectly are induced in the host immune cells (e.g., activation of immune suppressive cells). Therefore, deeper insights into the immuno-biology of CSCs are essential in our pursuit to find new therapeutic opportunities that eradicate cancer (stem) cells. Here, we review and discuss the ability of CSCs to evade the innate and adaptive immune system, as we offer a view of the immunotherapeutic strategies adopted to potentiate and address specific subsets of (engineered) immune cells against CSCs.
Keywords: cancer stem cells; immune escape; immunogenicity; immunotherapy; tumor microenvironment.
Conflict of interest statement
The authors have no relevant conflict of interest to declare.
Figures
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

