Lipid Polymorphism of the Subchloroplast-Granum and Stroma Thylakoid Membrane-Particles. II. Structure and Functions
- PMID: 34572012
- PMCID: PMC8472583
- DOI: 10.3390/cells10092363
Lipid Polymorphism of the Subchloroplast-Granum and Stroma Thylakoid Membrane-Particles. II. Structure and Functions
Abstract
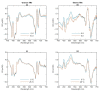
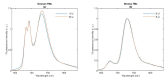
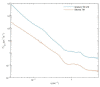
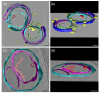
In Part I, by using 31P-NMR spectroscopy, we have shown that isolated granum and stroma thylakoid membranes (TMs), in addition to the bilayer, display two isotropic phases and an inverted hexagonal (HII) phase; saturation transfer experiments and selective effects of lipase and thermal treatments have shown that these phases arise from distinct, yet interconnectable structural entities. To obtain information on the functional roles and origin of the different lipid phases, here we performed spectroscopic measurements and inspected the ultrastructure of these TM fragments. Circular dichroism, 77 K fluorescence emission spectroscopy, and variable chlorophyll-a fluorescence measurements revealed only minor lipase- or thermally induced changes in the photosynthetic machinery. Electrochromic absorbance transients showed that the TM fragments were re-sealed, and the vesicles largely retained their impermeabilities after lipase treatments-in line with the low susceptibility of the bilayer against the same treatment, as reflected by our 31P-NMR spectroscopy. Signatures of HII-phase could not be discerned with small-angle X-ray scattering-but traces of HII structures, without long-range order, were found by freeze-fracture electron microscopy (FF-EM) and cryo-electron tomography (CET). EM and CET images also revealed the presence of small vesicles and fusion of membrane particles, which might account for one of the isotropic phases. Interaction of VDE (violaxanthin de-epoxidase, detected by Western blot technique in both membrane fragments) with TM lipids might account for the other isotropic phase. In general, non-bilayer lipids are proposed to play role in the self-assembly of the highly organized yet dynamic TM network in chloroplasts.
Keywords: SAXS; bilayer; chlorophyll fluorescence; cryo-electron-tomography; electron microscopy; membrane energization; membrane networks; non-bilayer lipid phases; violaxanthin de-epoxidase.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
Lipid polymorphism of plant thylakoid membranes. The dynamic exchange model - facts and hypotheses.Physiol Plant. 2025 Mar-Apr;177(2):e70230. doi: 10.1111/ppl.70230. Physiol Plant. 2025. PMID: 40251902 Free PMC article. Review.
-
Structural Entities Associated with Different Lipid Phases of Plant Thylakoid Membranes-Selective Susceptibilities to Different Lipases and Proteases.Cells. 2022 Aug 28;11(17):2681. doi: 10.3390/cells11172681. Cells. 2022. PMID: 36078087 Free PMC article.
-
Lipid Polymorphism of the Subchloroplast-Granum and Stroma Thylakoid Membrane-Particles. I. 31P-NMR Spectroscopy.Cells. 2021 Sep 8;10(9):2354. doi: 10.3390/cells10092354. Cells. 2021. PMID: 34572003 Free PMC article.
-
Role of isotropic lipid phase in the fusion of photosystem II membranes.Photosynth Res. 2024 Aug;161(1-2):127-140. doi: 10.1007/s11120-024-01097-3. Epub 2024 Apr 25. Photosynth Res. 2024. PMID: 38662326 Free PMC article.
-
Structural and functional roles of non-bilayer lipid phases of chloroplast thylakoid membranes and mitochondrial inner membranes.Prog Lipid Res. 2022 Apr;86:101163. doi: 10.1016/j.plipres.2022.101163. Epub 2022 Mar 26. Prog Lipid Res. 2022. PMID: 35351472 Review.
Cited by
-
Lipid Phase Behaviour of the Curvature Region of Thylakoid Membranes of Spinacia oleracea.Physiol Plant. 2025 May-Jun;177(3):e70289. doi: 10.1111/ppl.70289. Physiol Plant. 2025. PMID: 40525547 Free PMC article.
-
Cationic Proteins Rich in Lysine Residue Trigger Formation of Non-bilayer Lipid Phases in Model and Biological Membranes: Biophysical Methods of Study.J Membr Biol. 2023 Dec;256(4-6):373-391. doi: 10.1007/s00232-023-00292-y. Epub 2023 Sep 21. J Membr Biol. 2023. PMID: 37735238 Review.
-
Lipid polymorphism of plant thylakoid membranes. The dynamic exchange model - facts and hypotheses.Physiol Plant. 2025 Mar-Apr;177(2):e70230. doi: 10.1111/ppl.70230. Physiol Plant. 2025. PMID: 40251902 Free PMC article. Review.
-
Spruce versus Arabidopsis: different strategies of photosynthetic acclimation to light intensity change.Photosynth Res. 2022 Oct;154(1):21-40. doi: 10.1007/s11120-022-00949-0. Epub 2022 Aug 18. Photosynth Res. 2022. PMID: 35980499
-
Structural Entities Associated with Different Lipid Phases of Plant Thylakoid Membranes-Selective Susceptibilities to Different Lipases and Proteases.Cells. 2022 Aug 28;11(17):2681. doi: 10.3390/cells11172681. Cells. 2022. PMID: 36078087 Free PMC article.
References
-
- Douce R., Joyard J. Biosynthesis of Thylakoid Membrane Lipids. In: Ort D.R., Yocum C.F., Heichel I.F., editors. Oxygenic Photosynthesis: The Light Reactions. Springer; Dordrecht, The Netherlands: 1996. pp. 69–101.
-
- Garab G., Ughy B., de Waard P., Akhtar P., Javornik U., Kotakis C., Šket P., Karlický V., Materová Z., Špunda V., et al. Lipid polymorphism in chloroplast thylakoid membranes—as revealed by P-31-NMR and timeresolved merocyanine fluorescence spectroscopy. Sci. Rep. 2017;7 doi: 10.1038/s41598-017-13574-y. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
- 19-13637S/Grantová Agentura České Republiky
- K 128679/Hungarian Scientific Research Fund
- GINOP-2.3.2-15-2016-00058/National Research Development and Innovation Office of Hungary
- CZ.02.1.01/0.0/0.0/16_019/0000797/SustES - Adaptation strategies for sustainable ecosystem services and food security under adverse environmental conditions
- CZ.02.1.01/0.0/0.0/ 16_019/0000827/European Regional Development Fund
LinkOut - more resources
Full Text Sources

