Assessment of Antibiofilm Potencies of Nervonic and Oleic Acid against Acinetobacter baumannii Using In Vitro and Computational Approaches
- PMID: 34572317
- PMCID: PMC8466663
- DOI: 10.3390/biomedicines9091133
Assessment of Antibiofilm Potencies of Nervonic and Oleic Acid against Acinetobacter baumannii Using In Vitro and Computational Approaches
Abstract
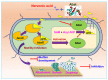
Acinetobacter baumannii is a nosocomial pathogen, and its biofilms are tolerant to desiccation, nutrient starvation, and antimicrobial treatment on biotic and abiotic surfaces, tissues, and medical devices. Biofilm formation by A. baumannii is triggered by a quorum sensing cascade, and we hypothesized that fatty acids might inhibit its biofilm formation by interfering with quorum sensing. Initially, we investigated the antibiofilm activities of 24 fatty acids against A. baumannii ATCC 17978 and two clinical isolates. Among these fatty acids, two unsaturated fatty acids, nervonic and oleic acid, at 20 μg/mL significantly inhibited A. baumannii biofilm formation without affecting its planktonic cell growth (MICs were >500 μg/mL) and markedly decreased the motility of A. baumannii but had no toxic effect on the nematode Caenorhabditis elegans. Interestingly, molecular dynamic simulations showed that both fatty acids bind to the quorum sensing acyl homoserine lactone synthase (AbaI), and decent conformational stabilities of interactions between the fatty acids and AbaI were exhibited. Our results demonstrate that nervonic and oleic acid inhibit biofilm formation by A. baumannii strains and may be used as lead molecules for the control of persistent A. baumannii infections.
Keywords: AbaI; Acinetobacter baumannii; antibiofilm agents; biofilm formation; computational studies; fatty acid; nervonic acid; quorum sensing; virulence.
Conflict of interest statement
The authors declare no conflict of interest.
Figures





Similar articles
-
Unsaturated Fatty Acids Affect Quorum Sensing Communication System and Inhibit Motility and Biofilm Formation of Acinetobacter baumannii.Int J Mol Sci. 2018 Jan 10;19(1):214. doi: 10.3390/ijms19010214. Int J Mol Sci. 2018. PMID: 29320462 Free PMC article.
-
Quorum Sensing as a Target for Controlling Surface Associated Motility and Biofilm Formation in Acinetobacter baumannii ATCC® 17978TM.Front Microbiol. 2020 Sep 30;11:565548. doi: 10.3389/fmicb.2020.565548. eCollection 2020. Front Microbiol. 2020. PMID: 33101239 Free PMC article.
-
Inhibition of Staphylococcus aureus Biofilm Formation and Virulence Factor Production by Petroselinic Acid and Other Unsaturated C18 Fatty Acids.Microbiol Spectr. 2022 Jun 29;10(3):e0133022. doi: 10.1128/spectrum.01330-22. Epub 2022 Jun 1. Microbiol Spectr. 2022. PMID: 35647620 Free PMC article.
-
Quorum-sensing system in Acinetobacter baumannii: a potential target for new drug development.J Appl Microbiol. 2020 Jan;128(1):15-27. doi: 10.1111/jam.14330. Epub 2019 Jun 26. J Appl Microbiol. 2020. PMID: 31102552 Review.
-
Quorum sensing in Acinetobacter: an emerging pathogen.Crit Rev Microbiol. 2010 Nov;36(4):349-60. doi: 10.3109/1040841X.2010.512269. Crit Rev Microbiol. 2010. PMID: 20846031 Review.
Cited by
-
Ice nucleation active bacteria metabolites as antibiofilm agent to control Aeromonas hydrophila and Streptococcus agalactiae infections in Aquaculture.BMC Res Notes. 2024 Jun 17;17(1):166. doi: 10.1186/s13104-024-06821-9. BMC Res Notes. 2024. PMID: 38886828 Free PMC article.
-
Motility of Acinetobacter baumannii: regulatory systems and controlling strategies.Appl Microbiol Biotechnol. 2024 Dec;108(1):3. doi: 10.1007/s00253-023-12975-6. Epub 2023 Dec 30. Appl Microbiol Biotechnol. 2024. PMID: 38159120 Review.
-
Exploring the potential of underrated yet versatile crop Lunaria annua L.: New insights into honesty plant.Heliyon. 2025 Jan 24;11(3):e42248. doi: 10.1016/j.heliyon.2025.e42248. eCollection 2025 Feb 15. Heliyon. 2025. PMID: 39944345 Free PMC article.
-
Exploring the dual effect of novel 1,4-diarylpyranopyrazoles as antiviral and anti-inflammatory for the management of SARS-CoV-2 and associated inflammatory symptoms.Bioorg Chem. 2023 Jan;130:106255. doi: 10.1016/j.bioorg.2022.106255. Epub 2022 Nov 17. Bioorg Chem. 2023. PMID: 36403336 Free PMC article.
-
Antimicrobial activity of D-amino acid in combination with photo-sonoactivated hypericin nanoparticles against Acinetobacter baumannii.BMC Microbiol. 2023 Jan 19;23(1):23. doi: 10.1186/s12866-023-02758-4. BMC Microbiol. 2023. PMID: 36658487 Free PMC article.
References
-
- Rumbo-Feal S., Gomez M.J., Gayoso C., Alvarez-Fraga L., Cabral M.P., Aransay A.M., Rodriguez-Ezpeleta N., Fullaondo A., Valle J., Tomas M., et al. Whole transcriptome analysis of Acinetobacter baumannii assessed by RNA-sequencing reveals different mRNA expression profiles in biofilm compared to planktonic cells. PLoS ONE. 2013;8:e72968. doi: 10.1371/journal.pone.0072968. - DOI - PMC - PubMed
-
- Eder A.E., Munir S.A., Hobby C.R., Anderson D.M., Herndon J.L., Siv A.W., Symes S.J.K., Giles D.K. Exogenous polyunsaturated fatty acids (PUFAs) alter phospholipid composition, membrane permeability, biofilm formation and motility in Acinetobacter baumannii. Microbiology. 2017;163:1626–1636. doi: 10.1099/mic.0.000556. - DOI - PubMed
-
- Kroger C., MacKenzie K.D., Alshabib E.Y., Kirzinger M.W.B., Suchan D.M., Chao T.C., Akulova V., Miranda-CasoLuengo A.A., Monzon V.A., Conway T., et al. The primary transcriptome, small RNAs and regulation of antimicrobial resistance in Acinetobacter baumannii ATCC 17978. Nucleic Acids Res. 2018;46:9684–9698. doi: 10.1093/nar/gky603. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

