Facile Generation of Potent Bispecific Fab via Sortase A and Click Chemistry for Cancer Immunotherapy
- PMID: 34572769
- PMCID: PMC8467688
- DOI: 10.3390/cancers13184540
Facile Generation of Potent Bispecific Fab via Sortase A and Click Chemistry for Cancer Immunotherapy
Abstract
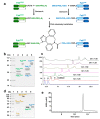
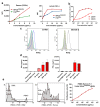
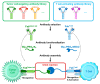
Bispecific antibodies (BsAbs) for T cell engagement have shown great promise in cancer immunotherapy, and their clinical applications have been proven in treating hematological malignance. Bispecific antibody binding fragment (BiFab) represents a promising platform for generating non-Fc bispecific antibodies. However, the generation of BiFab is still challenging, especially by means of chemical conjugation. More conjugation strategies, e.g., enzymatic conjugation and modular BiFab preparation, are needed to improve the robustness and flexibility of BiFab preparation. We successfully used chemo-enzymatic conjugation approach to generate bispecific antibody (i.e., BiFab) with Fabs from full-length antibodies. Paired click handles (e.g., N3 and DBCO) was introduced to the C-terminal LPETG tag of Fabs via sortase A mediated transpeptidation, followed by site-specific conjugation between two click handle-modified Fabs for BiFab generation. Both BiFabCD20/CD3 (EC50 = 0.26 ng/mL) and BiFabHer2/CD3 exhibited superior efficacy in mediating T cells, from either PBMC or ATC, to kill target tumor cell lines while spared antigen-negative tumor cells in vitro. The BiFabCD20/CD3 also efficiently inhibited CD20-positive tumor growth in mouse xenograft model. We have established a facile sortase A-mediated click handle installation to generate homogeneous and functional BiFabs. The exemplary BiFabs against different targets showed superior efficacy in redirecting and activating T cells to specifically kill target tumor cells, demonstrating the robustness of sortase A-mediated "bio-click" chemistry in generating various potent BiFabs. This approach also holds promise for further efficient construction of a Fab derivative library for personalized tumor immunotherapy in the future.
Keywords: BiFab; Fab; anti-CD20 antibody; bispecific antibody; chemo-enzymatic approach; sortase A.
Conflict of interest statement
The authors have filed a patent for the BiFab generation. Wenhui Liu is an employee of Hangzhou Biosun Pharmaceutical Co., Ltd.
Figures

Similar articles
-
Synthesis of Multiple Bispecific Antibody Formats with Only One Single Enzyme Based on Enhanced Trypsiligase.Int J Mol Sci. 2022 Mar 15;23(6):3144. doi: 10.3390/ijms23063144. Int J Mol Sci. 2022. PMID: 35328563 Free PMC article.
-
Preparation of bispecific antibody-protein adducts by site-specific chemo-enzymatic conjugation.Methods. 2019 Feb 1;154:93-101. doi: 10.1016/j.ymeth.2018.07.013. Epub 2018 Aug 3. Methods. 2019. PMID: 30081077
-
An anti-B cell maturation antigen bispecific antibody for multiple myeloma.J Am Chem Soc. 2015 Apr 29;137(16):5288-91. doi: 10.1021/jacs.5b01876. Epub 2015 Apr 15. J Am Chem Soc. 2015. PMID: 25826669
-
Bispecific antibodies and trispecific immunocytokines for targeting the immune system against cancer: preparing for the future.BioDrugs. 2013 Feb;27(1):35-53. doi: 10.1007/s40259-012-0008-z. BioDrugs. 2013. PMID: 23329400 Review.
-
Bispecific antibodies for the treatment of lymphomas: Promises and challenges.Hematol Oncol. 2021 Jun;39 Suppl 1:113-116. doi: 10.1002/hon.2858. Hematol Oncol. 2021. PMID: 34105818 Review.
Cited by
-
Leveraging immunoliposomes as nanocarriers against SARS-CoV-2 and its emerging variants.Asian J Pharm Sci. 2023 Nov;18(6):100855. doi: 10.1016/j.ajps.2023.100855. Epub 2023 Oct 20. Asian J Pharm Sci. 2023. PMID: 38125653 Free PMC article. Review.
-
Antibody-drug conjugates and bispecific antibodies targeting cancers: applications of click chemistry.Arch Pharm Res. 2023 Mar;46(3):131-148. doi: 10.1007/s12272-023-01433-6. Epub 2023 Mar 6. Arch Pharm Res. 2023. PMID: 36877356 Review.
-
Cancer Immunology and Immunotherapies: Mechanisms That Affect Antitumor Immune Response and Treatment Resistance.Cancers (Basel). 2021 Nov 12;13(22):5655. doi: 10.3390/cancers13225655. Cancers (Basel). 2021. PMID: 34830808 Free PMC article.
References
-
- Slaney C.Y., Wang P., Darcy P.K., Kershaw M.H. CARs versus BiTEs: A Comparison between T Cell–Redirection Strategies for Cancer Treatment. Cancer Discov. 2018;8:924–934. doi: 10.1158/2159-8290.CD-18-0297. - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

