Intermittent Theta Burst Stimulation to the Primary Motor Cortex Reduces Cortical Inhibition: A TMS-EEG Study
- PMID: 34573136
- PMCID: PMC8472376
- DOI: 10.3390/brainsci11091114
Intermittent Theta Burst Stimulation to the Primary Motor Cortex Reduces Cortical Inhibition: A TMS-EEG Study
Abstract
Introduction: The aim of this study was to reveal the effects of intermittent theta burst stimulation (iTBS) in modulating cortical networks using transcranial magnetic stimulation and electroencephalography (TMS-EEG) recording.
Methods: Eighteen young adults participated in our study and received iTBS to the primary motor cortex (M1), supplementary motor area, and the primary visual cortex in three separate sessions. A finger tapping task and ipsilateral single-pulse TMS-EEG recording for the M1 were administrated before and after iTBS in each session. The effects of iTBS in motor performance and TMS-evoked potentials (TEPs) were investigated.
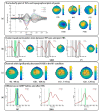
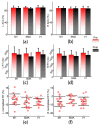
Results: The results showed that iTBS to the M1, but not supplementary motor area or the primary visual cortex, significantly reduced the N100 amplitude of M1 TEPs in bilateral hemispheres (p = 0.019), with a more prominent effect in the contralateral hemisphere than in the stimulated hemisphere. Moreover, only iTBS to the M1 decreased global mean field power (corrected ps < 0.05), interhemispheric signal propagation (t = 2.53, p = 0.030), and TMS-induced early α-band synchronization (p = 0.020).
Conclusion: Our study confirmed the local and remote after-effects of iTBS in reducing cortical inhibition in the M1. TMS-induced oscillations after iTBS for changed cortical excitability in patients with various neurological and psychiatric conditions are worth further exploration.
Keywords: TMS-evoked potentials; cortical inhibition; primary motor cortex; transcranial magnetic stimulation.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




References
-
- Carson R.G., Ruddy K.L., McNickle E. What do TMS-evoked motor potentials tell us about motor learning? Adv. Exp. Med. Biol. 2016;957:143–157. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

