pH-Responsive Alginate-Based Microparticles for Colon-Targeted Delivery of Pure Cyclosporine A Crystals to Treat Ulcerative Colitis
- PMID: 34575488
- PMCID: PMC8469027
- DOI: 10.3390/pharmaceutics13091412
pH-Responsive Alginate-Based Microparticles for Colon-Targeted Delivery of Pure Cyclosporine A Crystals to Treat Ulcerative Colitis
Abstract
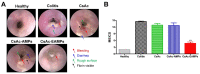
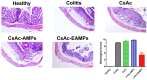
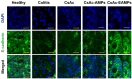
Cyclosporine A (CsA) is a potent immunosuppressant for treating ulcerative colitis (UC). However, owing to severe systemic side effects, CsA application in UC therapy remains limited. Herein, a colon-targeted drug delivery system consisting of CsA crystals (CsAc)-loaded, Eudragit S 100 (ES)-coated alginate microparticles (CsAc-EAMPs) was established to minimize systemic side effects and enhance the therapeutic efficacy of CsA. Homogeneously-sized CsAs (3.1 ± 0.9 μm) were prepared by anti-solvent precipitation, followed by the fabrication of 47.1 ± 6.5 μm-sized CsAc-EAMPs via ionic gelation and ES coating. CsAc-EAMPs exhibited a high drug loading capacity (48 ± 5%) and a CsA encapsulation efficacy of 77 ± 9%. The in vitro drug release study revealed that CsA release from CsAc-EAMPs was suppressed under conditions simulating the stomach and small intestine, resulting in minimized systemic absorption and side effects. Following exposure to the simulated colon conditions, along with ES dissolution and disintegration of alginate microparticles, CsA was released from CsAc-EAMPs, exhibiting a sustained-release profile for up to 24 h after administration. Given the effective colonic delivery of CsA molecules, CsAc-EAMPs conferred enhanced anti-inflammatory activity in mouse model of dextran sulfate sodium (DSS)-induced colitis. These findings suggest that CsAc-EAMPs is a promising drug delivery system for treating UC.
Keywords: colon-targeted delivery; cyclosporine A; ionic gelation; microparticles; ulcerative colitis.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




Similar articles
-
Alginate-Based Encapsulation Fabrication Technique for Drug Delivery: An Updated Review of Particle Type, Formulation Technique, Pharmaceutical Ingredient, and Targeted Delivery System.Pharmaceutics. 2024 Mar 6;16(3):370. doi: 10.3390/pharmaceutics16030370. Pharmaceutics. 2024. PMID: 38543264 Free PMC article. Review.
-
Colon-targeted delivery of cyclosporine A using dual-functional Eudragit® FS30D/PLGA nanoparticles ameliorates murine experimental colitis.Int J Nanomedicine. 2018 Feb 28;13:1225-1240. doi: 10.2147/IJN.S157566. eCollection 2018. Int J Nanomedicine. 2018. PMID: 29535519 Free PMC article.
-
Cupriavidus necator-Produced Polyhydroxybutyrate/Eudragit FS Hybrid Nanoparticles Mitigates Ulcerative Colitis via Colon-Targeted Delivery of Cyclosporine A.Pharmaceutics. 2022 Dec 15;14(12):2811. doi: 10.3390/pharmaceutics14122811. Pharmaceutics. 2022. PMID: 36559305 Free PMC article.
-
Dextran sulfate sodium-induced colitis and ginseng intervention altered oral pharmacokinetics of cyclosporine A in rats.J Ethnopharmacol. 2021 Jan 30;265:113251. doi: 10.1016/j.jep.2020.113251. Epub 2020 Aug 15. J Ethnopharmacol. 2021. PMID: 32810615
-
Pharmaceutical approaches to colon targeted drug delivery systems.J Pharm Pharm Sci. 2003 Jan-Apr;6(1):33-66. J Pharm Pharm Sci. 2003. PMID: 12753729 Review.
Cited by
-
pH-Dependent Drug Delivery Systems for Ulcerative Colitis Treatment.Pharmaceutics. 2025 Feb 10;17(2):226. doi: 10.3390/pharmaceutics17020226. Pharmaceutics. 2025. PMID: 40006593 Free PMC article. Review.
-
Alginate-Based Encapsulation Fabrication Technique for Drug Delivery: An Updated Review of Particle Type, Formulation Technique, Pharmaceutical Ingredient, and Targeted Delivery System.Pharmaceutics. 2024 Mar 6;16(3):370. doi: 10.3390/pharmaceutics16030370. Pharmaceutics. 2024. PMID: 38543264 Free PMC article. Review.
-
Mucoadhesive Nanoparticles Enhance the Therapeutic Effect of Dexamethasone on Experimental Ulcerative Colitis by the Local Administration as an Enema.Drug Des Devel Ther. 2023 Jan 24;17:191-207. doi: 10.2147/DDDT.S390274. eCollection 2023. Drug Des Devel Ther. 2023. PMID: 36718245 Free PMC article.
-
Potential of Nature-derived Biopolymers for Oral Applications- A Mini-Review.Mini Rev Med Chem. 2025;25(7):529-538. doi: 10.2174/0113895575359305241218113847. Mini Rev Med Chem. 2025. PMID: 39757671 Review.
-
Designing Advanced Drug Delivery Systems: Core-Shell Alginate Particles through Electro-Fluid Dynamic Atomization.Pharmaceutics. 2024 Jan 29;16(2):193. doi: 10.3390/pharmaceutics16020193. Pharmaceutics. 2024. PMID: 38399251 Free PMC article.
References
-
- Jeong S., Ju S., Park S., Jung Y. 5-[(3-Carboxy-4-hydroxyphenyl) diazenyl] nicotinic acid, an azo-linked mesalazine-nicotinic acid conjugate, is a colon-targeted mutual prodrug against dextran sulfate sodium-induced colitis in mice. J. Pharm. Investig. 2021;51:317–325. doi: 10.1007/s40005-021-00517-z. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources

