Identification of Clinical Isolates of Candida albicans with Increased Fitness in Colonization of the Murine Gut
- PMID: 34575733
- PMCID: PMC8468482
- DOI: 10.3390/jof7090695
Identification of Clinical Isolates of Candida albicans with Increased Fitness in Colonization of the Murine Gut
Abstract
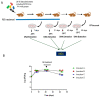
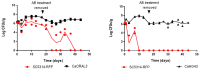
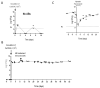
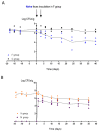
The commensal and opportunistic pathogen Candida albicans is an important cause of fungal diseases in humans, with the gastrointestinal tract being an important reservoir for its infections. The study of the mechanisms promoting the C. albicans commensal state has attracted considerable attention over the last few years, and several studies have focused on the identification of the intestinal human mycobiota and the characterization of Candida genes involved in its establishment as a commensal. In this work, we have barcoded 114 clinical C. albicans isolates to identify strains with an enhanced fitness in a murine gastrointestinal commensalism model. The 114 barcoded clinical isolates were pooled in four groups of 28 to 30 strains that were inoculated by gavage in mice previously treated with antibacterial therapy. Eight strains that either exhibited higher colonization load and/or remained in the gut after antibiotic removal were selected. The phenotypic analysis of these strains compared to an RFP-tagged SC5314 wild type strain did not reveal any specific trait associated with its increased colonization; all strains were able to filament and six of the eight strains displayed invasive growth on Spider medium. Analysis of one of these strains, CaORAL3, revealed that although mice required previous bacterial microbiota reduction with antibiotics to be able to be colonized, removal of this procedure could take place the same day (or even before) Candida inoculation. This strain was able to colonize the intestine of mice already colonized with Candida without antibiotic treatment in co-housing experiments. CaORAL3 was also able to be established as a commensal in mice previously colonized by another (CaHG43) or the same (CaORAL3) C. albicans strain. Therefore, we have identified C. albicans isolates that display higher colonization load than the standard strain SC5314 which will surely facilitate the analysis of the factors that regulate fungal colonization.
Keywords: Candida albicans; adaptation; commensalism; fitness; murine gut; mycobiota.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
-
- Odds F.C. Candida and Candidosis. Volume 2 Baillière Tindall; London, UK: 1988.
Grants and funding
LinkOut - more resources
Full Text Sources

