Evaluating Neuroprotective Effects of Uridine, Erythropoietin, and Therapeutic Hypothermia in a Ferret Model of Inflammation-Sensitized Hypoxic-Ischemic Encephalopathy
- PMID: 34576001
- PMCID: PMC8469346
- DOI: 10.3390/ijms22189841
Evaluating Neuroprotective Effects of Uridine, Erythropoietin, and Therapeutic Hypothermia in a Ferret Model of Inflammation-Sensitized Hypoxic-Ischemic Encephalopathy
Abstract
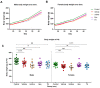
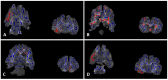
Perinatal hypoxic-ischemic (HI) brain injury, often in conjunction with an inflammatory insult, is the most common cause of death or disability in neonates. Therapeutic hypothermia (TH) is the standard of care for HI encephalopathy in term and near-term infants. However, TH may not always be available or efficacious, creating a need for novel or adjunctive neurotherapeutics. Using a near-term model of inflammation-sensitized HI brain injury in postnatal day (P) 17 ferrets, animals were randomized to either the control group (n = 43) or the HI-exposed groups: saline vehicle (Veh; n = 42), Ur (uridine monophosphate, n = 23), Epo (erythropoietin, n = 26), or TH (n = 24) to test their respective therapeutic effects. Motor development was assessed from P21 to P42 followed by analysis of cortical anatomy, ex vivo MRI, and neuropathology. HI animals took longer to complete the motor assessments compared to controls, which was exacerbated in the Ur group. Injury resulted in thinned white matter tracts and narrowed cortical sulci and gyri, which was mitigated in Epo-treated animals in addition to normalization of cortical neuropathology scores to control levels. TH and Epo treatment also resulted in region-specific improvements in diffusion parameters on ex vivo MRI; however, TH was not robustly neuroprotective in any behavioral or neuropathological outcome measures. Overall, Ur and TH did not provide meaningful neuroprotection after inflammation-sensitized HI brain injury in the ferret, and Ur appeared to worsen outcomes. By comparison, Epo appears to provide significant, though not complete, neuroprotection in this model.
Keywords: asphyxia; erythropoietin; neonatal; therapeutic hypothermia (TH); uridine.
Conflict of interest statement
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Figures




References
-
- Lee A.C., Kozuki N., Blencowe H., Vos T., Bahalim A., Darmstadt G.L., Niermeyer S., Ellis M., Robertson N.J., Cousens S., et al. Intrapartum-related neonatal encephalopathy incidence and impairment at regional and global levels for 2010 with trends from 1990. Pediatr. Res. 2013;74((Suppl. 1)):50–72. doi: 10.1038/pr.2013.206. - DOI - PMC - PubMed
-
- Manuck T.A., Rice M.M., Bailit J.L., Grobman W.A., Reddy U.M., Wapner R.J., Thorp J.M., Caritis S.N., Prasad M., Tita A.T., et al. Preterm neonatal morbidity and mortality by gestational age: A contemporary cohort. Am. J. Obstet. Gynecol. 2016;215:103.e1–103.e14. doi: 10.1016/j.ajog.2016.01.004. - DOI - PMC - PubMed
-
- Thayyil S., Pant S., Montaldo P., Shukla D., Oliveira V., Ivain P., Bassett P., Swamy R., Mendoza J., Moreno-Morales M., et al. Hypothermia for moderate or severe neonatal encephalopathy in low-income and middle-income countries (HELIX): A randomised controlled trial in India, Sri Lanka, and Bangladesh. Lancet Glob. Health. 2021;9:e1273–e1285. doi: 10.1016/S2214-109X(21)00264-3. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

