Sulfonium Ligands of the α7 nAChR
- PMID: 34577114
- PMCID: PMC8464850
- DOI: 10.3390/molecules26185643
Sulfonium Ligands of the α7 nAChR
Abstract
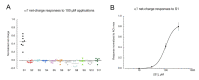
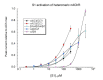
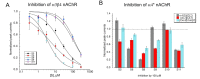
The α7 nicotinic acetylcholine receptor (nAChR) is an important target given its role in cognitive function as well as in the cholinergic anti-inflammatory pathway, where ligands that are effective at stabilizing desensitized states of the receptor are of particular interest. The typical structural element associated with a good desensitizer is the ammonium pharmacophore, but recent work has identified that a trivalent sulfur, in the positively charged sulfonium form, can substitute for the nitrogen in the ammonium pharmacophore. However, the breadth and scope of employing the sulfonium group is largely unexplored. In this work, we have surveyed a disparate group of sulfonium compounds for their functional activity with α7 as well as other nAChR subtypes. Amongst them, we found that there is a wide range of ability to induce α7 desensitization, with 4-hydroxyphenyldimethylsulfonium and suplatast sulfonium salts being the most desensitizing. The smallest sulfonium compound, trimethylsulfonium, was a partial agonist for α7 and other neuronal nAChR. Molecular docking into the α7 receptor extracellular domain revealed preferred poses in the orthosteric binding site for all but one compound, with typical cation-pi interactions as seen with traditional ammonium compounds. A number of the compounds tested may serve as useful platforms for further development of α7 desensitizing ability and for receptor subtype selectivity.
Keywords: desensitize; isostere; nicotinic acetylcholine receptor; silent agonist; sulfonium.
Conflict of interest statement
The authors declare no conflict of interest.
Figures





Similar articles
-
Sulfonium as a Surrogate for Ammonium: A New α7 Nicotinic Acetylcholine Receptor Partial Agonist with Desensitizing Activity.J Med Chem. 2017 Sep 28;60(18):7928-7934. doi: 10.1021/acs.jmedchem.7b00875. Epub 2017 Sep 14. J Med Chem. 2017. PMID: 28885019 Free PMC article.
-
Sulfonium Moieties as Ammonium Bioisosteres: Novel Ligands for the Alpha7 Nicotinic Acetylcholine Receptor.J Med Chem. 2025 Feb 13;68(3):3157-3179. doi: 10.1021/acs.jmedchem.4c02399. Epub 2025 Jan 20. J Med Chem. 2025. PMID: 39829331
-
Multiple interaction regions in the orthosteric ligand binding domain of the α7 neuronal nicotinic acetylcholine receptor.J Chem Inf Model. 2012 Nov 26;52(11):3064-73. doi: 10.1021/ci3001953. Epub 2012 Nov 6. J Chem Inf Model. 2012. PMID: 23092444
-
Therapeutic Targeting of α7 Nicotinic Acetylcholine Receptors.Pharmacol Rev. 2021 Jul;73(3):1118-1149. doi: 10.1124/pharmrev.120.000097. Pharmacol Rev. 2021. PMID: 34301823 Free PMC article. Review.
-
In Silico Modeling of the α7 Nicotinic Acetylcholine Receptor: New Pharmacological Challenges Associated with Multiple Modes of Signaling.Mini Rev Med Chem. 2020;20(10):841-864. doi: 10.2174/1389557520666200130105256. Mini Rev Med Chem. 2020. PMID: 32000651 Free PMC article. Review.
Cited by
-
Alkylated Sulfonium Modification of Low Molecular Weight Polyethylenimine to Form Lipopolymers as Gene Vectors.ACS Omega. 2024 Jan 4;9(2):2339-2349. doi: 10.1021/acsomega.3c06255. eCollection 2024 Jan 16. ACS Omega. 2024. PMID: 38250374 Free PMC article.
References
-
- Padgett C.L., Hanek A.P., Lester H.A., Dougherty D.A., Lummis S.C. Unnatural amino acid mutagenesis of the GABA(A) receptor binding site residues reveals a novel cation-pi interaction between GABA and beta 2Tyr97. J. Neurosci. 2007;27:886–892. doi: 10.1523/JNEUROSCI.4791-06.2007. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

