COVID-19 vaccination in kidney transplant recipients
- PMID: 34580488
- PMCID: PMC8475856
- DOI: 10.1038/s41581-021-00491-7
COVID-19 vaccination in kidney transplant recipients
Abstract
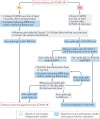
Kidney transplant recipients receive therapeutic immunosuppression that impairs their immune responses to the COVID-19 mRNA vaccine. For this reason, this vulnerable patient population is insufficiently protected by the standard two-dose COVID-19 vaccination programme and requires a specific follow-up to guide personalization of an intensified vaccination approach.
Conflict of interest statement
The authors declare no competing interests.
Figures
References
-
- Charmetant X, et al. Comparison of infected and vaccinated transplant recipients highlights the role of Tfh and neutralizing IgG in COVID-19 protection. medRxiv. 2021 doi: 10.1101/2021.07.22.21260852. - DOI
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

