Characteristics and Outcomes of Patients With Cerebral Venous Sinus Thrombosis in SARS-CoV-2 Vaccine-Induced Immune Thrombotic Thrombocytopenia
- PMID: 34581763
- PMCID: PMC8479648
- DOI: 10.1001/jamaneurol.2021.3619
Characteristics and Outcomes of Patients With Cerebral Venous Sinus Thrombosis in SARS-CoV-2 Vaccine-Induced Immune Thrombotic Thrombocytopenia
Abstract
Importance: Thrombosis with thrombocytopenia syndrome (TTS) has been reported after vaccination with the SARS-CoV-2 vaccines ChAdOx1 nCov-19 (Oxford-AstraZeneca) and Ad26.COV2.S (Janssen/Johnson & Johnson).
Objective: To describe the clinical characteristics and outcome of patients with cerebral venous sinus thrombosis (CVST) after SARS-CoV-2 vaccination with and without TTS.
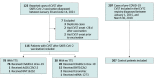
Design, setting, and participants: This cohort study used data from an international registry of consecutive patients with CVST within 28 days of SARS-CoV-2 vaccination included between March 29 and June 18, 2021, from 81 hospitals in 19 countries. For reference, data from patients with CVST between 2015 and 2018 were derived from an existing international registry. Clinical characteristics and mortality rate were described for adults with (1) CVST in the setting of SARS-CoV-2 vaccine-induced immune thrombotic thrombocytopenia, (2) CVST after SARS-CoV-2 vaccination not fulling criteria for TTS, and (3) CVST unrelated to SARS-CoV-2 vaccination.
Exposures: Patients were classified as having TTS if they had new-onset thrombocytopenia without recent exposure to heparin, in accordance with the Brighton Collaboration interim criteria.
Main outcomes and measures: Clinical characteristics and mortality rate.
Results: Of 116 patients with postvaccination CVST, 78 (67.2%) had TTS, of whom 76 had been vaccinated with ChAdOx1 nCov-19; 38 (32.8%) had no indication of TTS. The control group included 207 patients with CVST before the COVID-19 pandemic. A total of 63 of 78 (81%), 30 of 38 (79%), and 145 of 207 (70.0%) patients, respectively, were female, and the mean (SD) age was 45 (14), 55 (20), and 42 (16) years, respectively. Concomitant thromboembolism occurred in 25 of 70 patients (36%) in the TTS group, 2 of 35 (6%) in the no TTS group, and 10 of 206 (4.9%) in the control group, and in-hospital mortality rates were 47% (36 of 76; 95% CI, 37-58), 5% (2 of 37; 95% CI, 1-18), and 3.9% (8 of 207; 95% CI, 2.0-7.4), respectively. The mortality rate was 61% (14 of 23) among patients in the TTS group diagnosed before the condition garnered attention in the scientific community and 42% (22 of 53) among patients diagnosed later.
Conclusions and relevance: In this cohort study of patients with CVST, a distinct clinical profile and high mortality rate was observed in patients meeting criteria for TTS after SARS-CoV-2 vaccination.
Conflict of interest statement
Figures
References
-
- European Medicines Agency . Use of Vaxzevira to prevent COVID-19—article 5(3) procedure: assessment report. Accessed July 13, 2021. https://www.ema.europa.eu/en/documents/referral/use-vaxzevria-prevent-co...
-
- MacNeil JR, Su JR, Broder KR, et al. . Updated recommendations from the Advisory Committee on Immunization Practices for use of the Janssen (Johnson & Johnson) COVID-19 vaccine after reports of thrombosis with thrombocytopenia syndrome among vaccine recipients—United States, April 2021. MMWR Morb Mortal Wkly Rep. 2021;70(17):651-656. doi:10.15585/mmwr.mm7017e4 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous