Extracorporeal membrane oxygenation for COVID-19: evolving outcomes from the international Extracorporeal Life Support Organization Registry
- PMID: 34599878
- PMCID: PMC8480964
- DOI: 10.1016/S0140-6736(21)01960-7
Extracorporeal membrane oxygenation for COVID-19: evolving outcomes from the international Extracorporeal Life Support Organization Registry
Abstract
Background: Over the course of the COVID-19 pandemic, the care of patients with COVID-19 has changed and the use of extracorporeal membrane oxygenation (ECMO) has increased. We aimed to examine patient selection, treatments, outcomes, and ECMO centre characteristics over the course of the pandemic to date.
Methods: We retrospectively analysed the Extracorporeal Life Support Organization Registry and COVID-19 Addendum to compare three groups of ECMO-supported patients with COVID-19 (aged ≥16 years). At early-adopting centres-ie, those using ECMO support for COVID-19 throughout 2020-we compared patients who started ECMO on or before May 1, 2020 (group A1), and between May 2 and Dec 31, 2020 (group A2). Late-adopting centres were those that provided ECMO for COVID-19 only after May 1, 2020 (group B). The primary outcome was in-hospital mortality in a time-to-event analysis assessed 90 days after ECMO initiation. A Cox proportional hazards model was fit to compare the patient and centre-level adjusted relative risk of mortality among the groups.
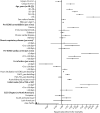
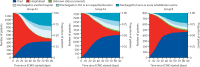
Findings: In 2020, 4812 patients with COVID-19 received ECMO across 349 centres within 41 countries. For early-adopting centres, the cumulative incidence of in-hospital mortality 90 days after ECMO initiation was 36·9% (95% CI 34·1-39·7) in patients who started ECMO on or before May 1 (group A1) versus 51·9% (50·0-53·8) after May 1 (group A2); at late-adopting centres (group B), it was 58·9% (55·4-62·3). Relative to patients in group A2, group A1 patients had a lower adjusted relative risk of in-hospital mortality 90 days after ECMO (hazard ratio 0·82 [0·70-0·96]), whereas group B patients had a higher adjusted relative risk (1·42 [1·17-1·73]).
Interpretation: Mortality after ECMO for patients with COVID-19 worsened during 2020. These findings inform the role of ECMO in COVID-19 for patients, clinicians, and policy makers.
Funding: None.
Copyright © 2021 Elsevier Ltd. All rights reserved.
Conflict of interest statement
Declaration of interests RPB is the ELSO Registry chair. GM, MLP, CMS, and DB are on the ELSO board of directors. MLP is the current president of ELSO and DB is the president-elect of ELSO. DB also chairs the executive committee for the International ECMO Network. PSB has received funding from ELSO for statistical analysis related and unrelated to this study. CMS receives payment from ELSO in her role as chief executive officer of ELSO. RPB, CA, and EF are members of the ELSO Steering Committee. RL and AC are past members of the European ELSO Steering Committee. ASS chairs the Scientific Oversight Committee of the International ECMO Network. RPB reports grants from the US National Institutes of Health (R01 HL153519, R01 HD015434, and K12 HL138039). AC reports speaking fees from Baxter, Getinge, and Fresenius, unrelated to the submitted work. EF reports consulting fees from ALung Technologies and Vasomune; speaking fees from Getinge; and fees for Data Safety Monitoring or Advisory Board participation from Baxter and Boehringer-Ingelheim. KH reports unpaid consulting work with Abbott Vascular. RL reports research support from Getinge; consulting fees from Getinge, Livanova, and Medtronic; and honoraria for lectures from Getinge and Livanova, unrelated to the submitted work. TJI is employed by the US Government. ASS reports consulting fees from Baxter and Xenios, unrelated to the submitted work. DB reports grants from ALung Technologies, and medical advisory board relationships with Baxter (past), Xenios, Abiomed, Cellenkos, Medtronic, and Hemovent, unrelated to the submitted work. All other authors declare no competing interests.
Figures

Comment in
-
ECMO in COVID-19: do not blame the tool.Lancet. 2021 Oct 2;398(10307):1197-1199. doi: 10.1016/S0140-6736(21)02137-1. Epub 2021 Sep 29. Lancet. 2021. PMID: 34599877 Free PMC article. No abstract available.
-
Extracorporeal membrane oxygenation for COVID-19: Some answers and a remaining question.Anaesth Crit Care Pain Med. 2022 Feb;41(1):100986. doi: 10.1016/j.accpm.2021.100986. Epub 2021 Nov 25. Anaesth Crit Care Pain Med. 2022. PMID: 34838733 Free PMC article. No abstract available.
-
COVID-19 ARDS: getting ventilation right.Lancet. 2022 Jan 1;399(10319):22. doi: 10.1016/S0140-6736(21)02439-9. Lancet. 2022. PMID: 34973714 Free PMC article. No abstract available.
References
-
- National Institutes of Health; COVID-19 Treatment Guidelines Panel Coronavirus disease 2019 (COVID-19) treatment guidelines. August 4, 2021. https://www.covid19treatmentguidelines.nih.gov/ - PubMed
-
- Combes A, Hajage D, Capellier G, et al. Extracorporeal membrane oxygenation for severe acute respiratory distress syndrome. N Engl J Med. 2018;378:1965–1975. - PubMed
-
- Peek GJ, Mugford M, Tiruvoipati R, et al. Efficacy and economic assessment of conventional ventilatory support versus extracorporeal membrane oxygenation for severe adult respiratory failure (CESAR): a multicentre randomised controlled trial. Lancet. 2009;374:1351–1363. - PubMed
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials

