Ancient Relatives of Modern Maize From the Center of Maize Domestication and Diversification Host Endophytic Bacteria That Confer Tolerance to Nitrogen Starvation
- PMID: 34603345
- PMCID: PMC8485183
- DOI: 10.3389/fpls.2021.660673
Ancient Relatives of Modern Maize From the Center of Maize Domestication and Diversification Host Endophytic Bacteria That Confer Tolerance to Nitrogen Starvation
Abstract
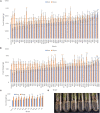
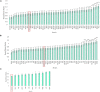
Plants can adapt to their surroundings by hosting beneficial bacteria that confer a selective advantage in stressful conditions. Endophytes are a class of beneficial bacteria that exist within the internal spaces of plants and many species can improve plant nitrogen use efficiency. Nitrogen is an essential plant macronutrient, and is often a limiting factor to plant growth, especially in cereal crops such as maize. Every year farmers apply over 100 million metric tonnes of synthetic nitrogen fertilizer to meet the growing demand for stable food crops. Breeding efforts in maize over the past several decades has focused heavily on yield in response to nitrogen inputs, and so may have selected against adaptations that allow plants to survive in nitrogen stressed conditions. Data suggests that our heavy dependence on synthetic nitrogen fertilizer is not sustainable in the long term, and so there is on-going research efforts to reduce and replace this currently essential part of modern agriculture. Bacteria that improve plant tolerance to nitrogen stressed environments would allow farmers to reduce the amount of fertilizer they apply. The selection of maize under high nitrogen conditions to create modern varieties may have caused the plant to lose these beneficial bacteria that allowed wild maize ancestors to thrive in low nitrogen soil. Here in this study, we examine the root and shoot microbiomes of the wild ancestor of all maize, Parviglumis, and an ancient Mexican landrace (Mixteco) from Oaxaca, the area of early maize diversification. Both of these maize genotypes have thrived for thousands of years with little to no nitrogen inputs and so we hypothesized that they host beneficial bacteria that allow them to thrive in nitrogen stressed conditions. We identified multiple root endophyte species from each ancient maize relative that increased the growth of annual ryegrass (model maize relative) under nitrogen starvation. Furthermore, research infers these strains were vertically transmitted to new generations of plants, potentially through seed, indicating selection pressure for Parviglumis and Mixteco to maintain them in their microbiome.
Keywords: diazotroph; domestication; endophyte; landrace; maize; microbiome; nitrogen; teosinte.
Copyright © 2021 Dumigan, Muileboom, Gregory, Shrestha, Hewedy and Raizada.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures



References
LinkOut - more resources
Full Text Sources
Miscellaneous

