Designing spatial and temporal control of vaccine responses
- PMID: 34603749
- PMCID: PMC8477997
- DOI: 10.1038/s41578-021-00372-2
Designing spatial and temporal control of vaccine responses
Abstract
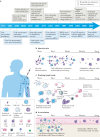
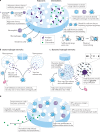
Vaccines are the key technology to combat existing and emerging infectious diseases. However, increasing the potency, quality and durability of the vaccine response remains a challenge. As our knowledge of the immune system deepens, it becomes clear that vaccine components must be in the right place at the right time to orchestrate a potent and durable response. Material platforms, such as nanoparticles, hydrogels and microneedles, can be engineered to spatially and temporally control the interactions of vaccine components with immune cells. Materials-based vaccination strategies can augment the immune response by improving innate immune cell activation, creating local inflammatory niches, targeting lymph node delivery and controlling the time frame of vaccine delivery, with the goal of inducing enhanced memory immunity to protect against future infections. In this Review, we highlight the biological mechanisms underlying strong humoral and cell-mediated immune responses and explore materials design strategies to manipulate and control these mechanisms.
Keywords: Biomaterials - vaccines; Biomedical engineering; Chemical engineering; Vaccines.
© Springer Nature Limited 2021.
Conflict of interest statement
Competing interestsG.A.R. and E.A.A. are listed as inventors on patent application describing sustained vaccine delivery (WO/2020/072495). The other authors declare no competing interests.
Figures




Similar articles
-
Nanovaccines for Advancing Long-Lasting Immunity against Infectious Diseases.ACS Nano. 2023 Dec 26;17(24):24514-24538. doi: 10.1021/acsnano.3c07741. Epub 2023 Dec 6. ACS Nano. 2023. PMID: 38055649 Review.
-
Modulation of injectable hydrogel properties for slow co-delivery of influenza subunit vaccine components enhance the potency of humoral immunity.J Biomed Mater Res A. 2021 Nov;109(11):2173-2186. doi: 10.1002/jbm.a.37203. Epub 2021 May 6. J Biomed Mater Res A. 2021. PMID: 33955657 Free PMC article.
-
Engineering Strategies for Lymph Node Targeted Immune Activation.Acc Chem Res. 2020 Oct 20;53(10):2055-2067. doi: 10.1021/acs.accounts.0c00260. Epub 2020 Sep 10. Acc Chem Res. 2020. PMID: 32910636
-
Optimize Prime/Boost Vaccine Strategies: Trained Immunity as a New Player in the Game.Front Immunol. 2021 Mar 8;12:612747. doi: 10.3389/fimmu.2021.612747. eCollection 2021. Front Immunol. 2021. PMID: 33763063 Free PMC article. Review.
-
Adjuvanting a Simian Immunodeficiency Virus Vaccine with Toll-Like Receptor Ligands Encapsulated in Nanoparticles Induces Persistent Antibody Responses and Enhanced Protection in TRIM5α Restrictive Macaques.J Virol. 2017 Jan 31;91(4):e01844-16. doi: 10.1128/JVI.01844-16. Print 2017 Feb 15. J Virol. 2017. PMID: 27928002 Free PMC article.
Cited by
-
A Biomimetic Multi-Component Subunit Vaccine via Ratiometric Loading of Hierarchical Hydrogels.Res Sq [Preprint]. 2024 Apr 25:rs.3.rs-4177821. doi: 10.21203/rs.3.rs-4177821/v1. Res Sq. 2024. Update in: Nat Commun. 2025 Jul 1;16(1):5443. doi: 10.1038/s41467-025-60416-x. PMID: 38746232 Free PMC article. Updated. Preprint.
-
Optimization of methods for isolation and purification of outer membrane vesicles (OMVs) from Neisseria lactamica.Appl Microbiol Biotechnol. 2025 Apr 7;109(1):82. doi: 10.1007/s00253-025-13460-y. Appl Microbiol Biotechnol. 2025. PMID: 40192813 Free PMC article.
-
In situ generation of micrometer-sized tumor cell-derived vesicles as autologous cancer vaccines for boosting systemic immune responses.Nat Commun. 2022 Nov 1;13(1):6534. doi: 10.1038/s41467-022-33831-7. Nat Commun. 2022. PMID: 36319625 Free PMC article.
-
A Novel Approach to Vaccine Development: Concomitant Pathogen Inactivation and Host Immune Stimulation by Peroxynitrite.Vaccines (Basel). 2022 Sep 22;10(10):1593. doi: 10.3390/vaccines10101593. Vaccines (Basel). 2022. PMID: 36298458 Free PMC article.
-
Injectable Liposome-based Supramolecular Hydrogels for the Programmable Release of Multiple Protein Drugs.Matter. 2022 Jun 1;5(6):1816-1838. doi: 10.1016/j.matt.2022.03.001. Epub 2022 Mar 21. Matter. 2022. PMID: 35800848 Free PMC article.
References
-
- World Health Organization. Global Vaccine Action Plan 2011–2020 (WHO, 2013).
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
