Rejigging Electron and Proton Transfer to Transition between Dioxygenase, Monooxygenase, Peroxygenase, and Oxygen Reduction Activity: Insights from Bioinspired Constructs of Heme Enzymes
- PMID: 34604840
- PMCID: PMC8479764
- DOI: 10.1021/jacsau.1c00100
Rejigging Electron and Proton Transfer to Transition between Dioxygenase, Monooxygenase, Peroxygenase, and Oxygen Reduction Activity: Insights from Bioinspired Constructs of Heme Enzymes
Abstract
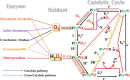
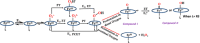
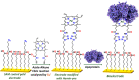
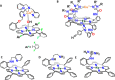
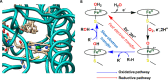
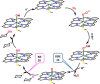
Nature has employed heme proteins to execute a diverse set of vital life processes. Years of research have been devoted to understanding the factors which bias these heme enzymes, with all having a heme cofactor, toward distinct catalytic activity. Among them, axial ligation, distal super structure, and substrate binding pockets are few very vividly recognized ones. Detailed mechanistic investigation of these heme enzymes suggested that several of these enzymes, while functionally divergent, use similar intermediates. Furthermore, the formation and decay of these intermediates depend on proton and electron transfer processes in the enzyme active site. Over the past decade, work in this group, using in situ surface enhanced resonance Raman spectroscopy of synthetic and biosynthetic analogues of heme enzymes, a general idea of how proton and electron transfer rates relate to the lifetime of different O2 derived intermediates has been developed. These findings suggest that the enzymatic activities of all these heme enzymes can be integrated into one general cycle which can be branched out to different catalytic pathways by regulating the lifetime and population of each of these intermediates. This regulation can further be achieved by tuning the electron and proton transfer steps. By strategically populating one of these intermediates during oxygen reduction, one can navigate through different catalytic processes to a desired direction by altering proton and electron transfer steps.
© 2021 The Authors. Published by American Chemical Society.
Conflict of interest statement
The authors declare no competing financial interest.
Figures




References
-
- Lee D.; Pierce B.; Krebs C.; Hendrich M. P.; Huynh B. H.; Lippard S. J. Functional Mimic of Dioxygen-Activating Centers in Non-Heme Diiron Enzymes: Mechanistic Implications of Paramagnetic Intermediates in the Reactions between Diiron(II) Complexes and Dioxygen. J. Am. Chem. Soc. 2002, 124, 3993–4007. 10.1021/ja012251t. - DOI - PubMed
-
- Collman J. P.; Fu L. Synthetic Models for Hemoglobin and Myoglobin. Acc. Chem. Res. 1999, 32, 455–463. 10.1021/ar9603064. - DOI
Publication types
LinkOut - more resources
Full Text Sources
Miscellaneous
