Serological response to nifurtimox in adult patients with chronic Chagas disease: An observational comparative study in Argentina
- PMID: 34606501
- PMCID: PMC8489720
- DOI: 10.1371/journal.pntd.0009801
Serological response to nifurtimox in adult patients with chronic Chagas disease: An observational comparative study in Argentina
Abstract
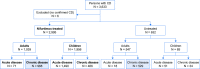
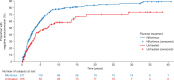
Nifurtimox is indicated in Chagas disease but determining its effectiveness in chronic disease is hindered by the length of time needed to demonstrate negative serological conversion. We manually reviewed long-term follow-up data from hospital records of patients with chronic Chagas disease (N = 1,497) in Argentina diagnosed during 1967-1980. All patients were aged ≥18 years at diagnosis and were either treated with nifurtimox (n = 968) or received no antitrypanosomal treatment (n = 529). The primary endpoint was negative seroconversion (the "event"), defined as a change from positive to negative in the serological or parasitological laboratory test used at diagnosis. Time to event was from baseline visit to date of endpoint event or censoring. The effectiveness of nifurtimox versus no treatment was estimated with Cox proportional hazard regression using propensity scores with overlap weights to calculate the hazard ratio and 95% confidence interval. The nifurtimox group was younger than the untreated group (mean, 32.4 vs. 40.3 years), with proportionally fewer females (47.9% vs. 60.1%), and proportionally more of the nifurtimox group than the untreated group had clinical signs and symptoms of Chagas disease at diagnosis (28.9% vs. 14.0%). Median maximum daily dose of nifurtimox was 8.0 mg/kg/day (interquartile range [IQR]: 8.0-9.0) and median treatment duration was 44 days (IQR: 1-90). Median time to event was 2.1 years (IQR: 1.0-4.5) for nifurtimox-treated and 2.4 years (IQR: 1.0-4.2) for untreated patients. Accounting for potential confounders, the estimated hazard ratio (95% confidence interval) for negative seroconversion was 2.22 (1.61-3.07) favoring nifurtimox. Variable treatment regimens and follow-up duration, and an uncommonly high rate of spontaneous negative seroconversion, complicate interpretation of this epidemiological study, but with the longest follow-up and largest cohort analyzed to date it lends weight to the benefit of nifurtimox in adults with chronic Chagas disease. Trial registration: The study protocol was registered at ClinicalTrials.gov: NCT03784391.
Conflict of interest statement
I have read the journal’s policy and the authors of this manuscript have the following competing interests: David Vizcaya, Ulrike Grossmann, Frank Kleinjung, Ruiping Zhang and Kiliana Suzart-Woischnik are employees of Bayer. Leylen Colmegna is an employee of LAT Research, Argentina. Sandra Seu, Teresa Ramirez and Oscar Ledesma have nothing to disclose.
Figures
Similar articles
-
Redefining the treatment of Chagas disease: a review of recent clinical and pharmacological data for a novel formulation of nifurtimox.PLoS Negl Trop Dis. 2025 Feb 25;19(2):e0012849. doi: 10.1371/journal.pntd.0012849. eCollection 2025 Feb. PLoS Negl Trop Dis. 2025. PMID: 39999088 Free PMC article. Review.
-
Quantification of Immunoglobulin G against Trypanosoma cruzi in Individuals with Chronic Chagas Disease Treated with Nifurtimox and Evaluated in Prolonged Follow-Up.Korean J Parasitol. 2019 Feb;57(1):39-41. doi: 10.3347/kjp.2019.57.1.39. Epub 2019 Feb 26. Korean J Parasitol. 2019. PMID: 30840798 Free PMC article.
-
Prospective, historically controlled study to evaluate the efficacy and safety of a new paediatric formulation of nifurtimox in children aged 0 to 17 years with Chagas disease one year after treatment (CHICO).PLoS Negl Trop Dis. 2021 Jan 7;15(1):e0008912. doi: 10.1371/journal.pntd.0008912. eCollection 2021 Jan. PLoS Negl Trop Dis. 2021. PMID: 33412557 Free PMC article. Clinical Trial.
-
Effectiveness of Nifurtimox in the Treatment of Chagas Disease: a Long-Term Retrospective Cohort Study in Children and Adults.Antimicrob Agents Chemother. 2022 May 17;66(5):e0202121. doi: 10.1128/aac.02021-21. Epub 2022 Apr 13. Antimicrob Agents Chemother. 2022. PMID: 35416710 Free PMC article.
-
The Chagas disease study landscape: A systematic review of clinical and observational antiparasitic treatment studies to assess the potential for establishing an individual participant-level data platform.PLoS Negl Trop Dis. 2021 Aug 16;15(8):e0009697. doi: 10.1371/journal.pntd.0009697. eCollection 2021 Aug. PLoS Negl Trop Dis. 2021. PMID: 34398888 Free PMC article.
Cited by
-
Triatoma dimidiata, domestic animals and acute Chagas disease: a 10-year follow-up after an eco-bio-social intervention.Parasit Vectors. 2025 Jul 4;18(1):253. doi: 10.1186/s13071-025-06897-7. Parasit Vectors. 2025. PMID: 40616155 Free PMC article.
-
Redefining the treatment of Chagas disease: a review of recent clinical and pharmacological data for a novel formulation of nifurtimox.PLoS Negl Trop Dis. 2025 Feb 25;19(2):e0012849. doi: 10.1371/journal.pntd.0012849. eCollection 2025 Feb. PLoS Negl Trop Dis. 2025. PMID: 39999088 Free PMC article. Review.
-
Triatoma dimidiata, domestic animals and acute Chagas disease: A 10 year follow-up after an eco-bio-social intervention.medRxiv [Preprint]. 2025 Mar 13:2025.03.11.25323671. doi: 10.1101/2025.03.11.25323671. medRxiv. 2025. Update in: Parasit Vectors. 2025 Jul 4;18(1):253. doi: 10.1186/s13071-025-06897-7. PMID: 40162261 Free PMC article. Updated. Preprint.
-
Drugs for Vector-Borne Protozoal Diseases in a One Health Scenario. A European Perspective.ACS Infect Dis. 2024 Nov 8;10(11):3715-3720. doi: 10.1021/acsinfecdis.4c00339. Epub 2024 Oct 29. ACS Infect Dis. 2024. PMID: 39471826 Free PMC article. Review.
-
Efficacy and Safety of Nifurtimox in Pediatric Patients with Chagas Disease: Results at 4-Year Follow-Up in a Prospective, Historically Controlled Study (CHICO SECURE).Antimicrob Agents Chemother. 2023 Apr 18;67(4):e0119322. doi: 10.1128/aac.01193-22. Epub 2023 Mar 28. Antimicrob Agents Chemother. 2023. PMID: 36975790 Free PMC article. Clinical Trial.
References
-
- World Health Organization. Chagas disease (also known as American trypanosomiasis). 2020 [Cited 2021 February 11]. Available from: https://www.who.int/en/news-room/fact-sheets/detail/chagas-disease-(amer....
Publication types
MeSH terms
Substances
Associated data
LinkOut - more resources
Full Text Sources
Medical

