Vanillin enones as selective inhibitors of the cancer associated carbonic anhydrase isoforms IX and XII. The out of the active site pocket for the design of selective inhibitors?
- PMID: 34607524
- PMCID: PMC8510595
- DOI: 10.1080/14756366.2021.1982933
Vanillin enones as selective inhibitors of the cancer associated carbonic anhydrase isoforms IX and XII. The out of the active site pocket for the design of selective inhibitors?
Abstract
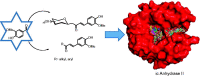
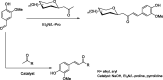
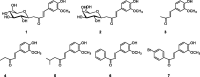
New C-glycosides and α,β-unsaturated ketones incorporating the 4-hydroxy-3-methoxyphenyl (vanillin) moiety as inhibitors of carbonic anhydrase (CA, EC 4.2.1.1) isoforms have been investigated. The inhibition profile of these compounds is presented against four human CA (hCA) isozymes, comprising hCAs I and II (cytosolic, ubiquitous enzymes) and hCAs IX and XII (tumour associated isozymes). Docking analysis of the inhibitors within the active sites of these enzymes has been performed and is discussed, showing that the observed selectivity could be explained in terms of an alternative pocket out of the CA active site where some of these compounds may bind. Several derivatives were identified as selective inhibitors of the tumour-associated hCA IX and XII. Their discovery might be a step in the strategy for finding an effective non-sulfonamide CA inhibitor useful in therapy/diagnosis of hypoxic tumours or other pathologies in which CA isoforms are involved.
Keywords: Vanillin; carbonic anhydrase; enones; enzyme inhibitors; molecular docking.
Conflict of interest statement
CT Supuran is Editor-in-Chief of Journal of Enzyme Inhibition and Medicinal Chemistry and he was not involved in the assessment, peer review, or decision-making process of this paper. The authors have no relevant affiliations or financial involvement with any organisation or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
Figures

Similar articles
-
S-substituted 2-mercaptoquinazolin-4(3H)-one and 4-ethylbenzensulfonamides act as potent and selective human carbonic anhydrase IX and XII inhibitors.J Enzyme Inhib Med Chem. 2020 Dec;35(1):733-743. doi: 10.1080/14756366.2020.1742117. J Enzyme Inhib Med Chem. 2020. PMID: 32189526 Free PMC article.
-
Novel benzenesulfonamide derivatives as potential selective carbonic anhydrase IX, XII inhibitors with anti-proliferative activity: Design, synthesis and in silico studies.Bioorg Chem. 2024 Dec;153:107881. doi: 10.1016/j.bioorg.2024.107881. Epub 2024 Oct 10. Bioorg Chem. 2024. PMID: 39396453
-
Design, synthesis and biological evaluation of coumarin-3-carboxamides as selective carbonic anhydrase IX and XII inhibitors.Bioorg Chem. 2019 May;86:386-392. doi: 10.1016/j.bioorg.2019.02.004. Epub 2019 Feb 5. Bioorg Chem. 2019. PMID: 30763885
-
Exploring the SAR of 1,2,3-Triazoles as Tumor-Associated Carbonic Anhydrases IX and XII Inhibitors for Anticancer Applications.Arch Pharm (Weinheim). 2025 Jul;358(7):e70041. doi: 10.1002/ardp.70041. Arch Pharm (Weinheim). 2025. PMID: 40629941 Review.
-
Antibody-drug conjugates and radioconjugates targeting carbonic anhydrase IX and XII in hypoxic tumors: Bench to clinical applications.Bioorg Chem. 2025 Jun 1;159:108408. doi: 10.1016/j.bioorg.2025.108408. Epub 2025 Mar 25. Bioorg Chem. 2025. PMID: 40154235 Review.
References
-
- a) Supuran CT. Carbonic anhydrases: novel therapeutic applications for inhibitors and activators. Nat Rev Drug Discov 2008;7:168–81.b) Supuran CT. Structure and function of carbonic anhydrases. Biochem J 2016;473: 2023,–32.c) Mishra CB, Tiwari M, Supuran CT.. Progress in the development of human carbonic anhydrase inhibitors and their pharmacological applications: where are we today? Med Res Rev 2020;40: 2485,–565.d) Supuran CT. Exploring the multiple binding modes of inhibitors to carbonic anhydrases for novel drug discovery. Expert Opin Drug Discov 2020;15: 671–86.
-
- a) Supuran CT. Emerging role of carbonic anhydrase inhibitors. Clin Sci 2021;135:1233–49.b) Supuran CT. Carbonic anhydrase inhibitors. Bioorg Med Chem Lett 2010;20: 3467,–74.c) Capasso C, Supuran CT.. An overview of the alpha-, beta- and gamma-carbonic anhydrases from Bacteria: can bacterial carbonic anhydrases shed new light on evolution of bacteria? J Enzyme Inhib Med Chem 2015;30: 325,–32.d) Supuran CT, Capasso C.. Antibacterial carbonic anhydrase inhibitors: an update on the recent literature. Expert Opin Ther Pat 2020;30: 963–82.
-
- a) Hirakawa Y, Senda M, Fukuda KY, et al. . Characterization of a novel type of carbonic anhydrase that acts without metal cofactors. BMC Biol 2021;19:105.b) Del Prete S, Nocentini A, Supuran CT, Capasso C.. Bacterial ι-carbonic anhydrase: a new active class of carbonic anhydrase identified in the genome of the Gram-negative bacterium Burkholderia territorii. J Enzyme Inhib Med Chem 2020;35: 1060,–8.c) Nocentini A, Supuran CT, Capasso C.. An overview on the recently discovered iota-carbonic anhydrases. J Enzyme Inhib Med Chem 2021;36: 1988–95.
-
- a) Pastorek J, Pastoreková S, Callebaut I, et al. . Cloning and characterization of MN, a human tumor-associated protein with a domain homologous to carbonic anhydrase and a putative helix-loop-helix DNA binding segment. Oncogene 1994;9:2877–88.b) Türeci O, Sahin U, Vollmar E, et al. . Human carbonic anhydrase XII: cDNA cloning, expression, and chromosomal localization of a carbonic anhydrase gene that is overexpressed in some renal cell cancers. Proc Natl Acad Sci USA 1998;95: 7608,–13.c) Svastová E, Hulíková A, Rafajová M, et al. . Hypoxia activates the capacity of tumor-associated carbonic anhydrase IX to acidify extracellular pH. FEBS Lett 2004;577: 439,–45.d) Supuran CT. Experimental carbonic anhydrase inhibitors for the treatment of hypoxic tumors. J Exp Pharmacol 2020;12: 603,–17.e) Supuran CT. Experimental carbonic anhydrase inhibitors for the treatment of hypoxic tumors. J Exp Pharmacol 2020;12: 603–17.
-
- a) Nocentini A, Angeli A, Carta F, et al. . Reconsidering anion inhibitors in the general context of drug design studies of modulators of activity of the classical enzyme carbonic anhydrase. J Enzyme Inhib Med Chem 2021;36:561–80.b) Angeli A, Carta F, Nocentini A, et al. . Carbonic anhydrase inhibitors targeting metabolism and tumor microenvironment. Metabolites 2020;10: 412,.c) McDonald PC, Chia S, Bedard PL, et al. . A phase 1 study of SLC-0111, a novel inhibitor of carbonic anhydrase IX, in patients with advanced solid tumors. Am J Clin Oncol 2020;43: 484,–90.d) Neri D, Supuran CT.. Interfering with pH regulation in tumours as a therapeutic strategy. Nat Rev Drug Discov 2011;10: 767,–77.e) Chafe SC, Vizeacoumar FS, Venkateswaran G, et al. . Genome-wide synthetic lethal screen unveils novel CAIX-NFS1/xCT axis as a targetable vulnerability in hypoxic solid tumors. Sci Adv 2021;7: eabj0364.
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
