Homebrewed psilocybin: can new routes for pharmaceutical psilocybin production enable recreational use?
- PMID: 34607532
- PMCID: PMC8806991
- DOI: 10.1080/21655979.2021.1987090
Homebrewed psilocybin: can new routes for pharmaceutical psilocybin production enable recreational use?
Abstract
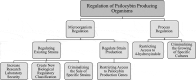
Psilocybin, a drug most commonly recognized as a recreational psychedelic, is quickly gaining attention as a promising therapy for an expanding range of neurological conditions, including depression, anxiety, and addiction. This growing interest has led to many recent advancements in psilocybin synthesis strategies, including multiple in vivo fermentation-based approaches catalyzed by recombinant microorganisms. In this work, we show that psilocybin can be produced in biologically relevant quantities using a recombinant E. coli strain in a homebrew style environment. In less than 2 days, we successfully produced approximately 300 mg/L of psilocybin under simple conditions with easily sourced equipment and supplies. This finding raises the question of how this new technology should be regulated as to not facilitate clandestine biosynthesis efforts, while still enabling advancements in psilocybin synthesis technology for pharmaceutical applications. Here, we present our homebrew results, and suggestions on how to address the regulatory concerns accompanying this new technology.
Keywords: Psilocybin; homebrewed drugs; recombinant DNA technology; regulation.
Conflict of interest statement
JAJ is the chairman of the scientific advisory board and a significant stakeholder at PsyBio Therapeutics. PsyBio Therapeutics has licensed psilocybin biosynthesis-related technology from Miami University. JAJ and WJGJ are co-inventors on a related provisional patent application.
Figures

References
-
- Gadsby R. Insulin treatment in diabetes. InnovAiT. 2013;6(6):344–348.
-
- Manco-Johnson MJ. Advances in the care and treatment of children with hemophilia. Adv Pediatr. 2010;57(1):287–294. - PubMed
-
- Paoletti MG, Pimentel D. Genetic engineering in agriculture and the environment: assessing risks and benefits. Bioscience. 1996;46(9):665–673.
-
- O’Sullivan M, Fox PF. Evaluation of microbial chymosin from genetically engineered kluyveromyces lactis. Food Biotechnol. 1991;5(1):19–32.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
