Building and Maintaining the Skin
- PMID: 34607830
- PMCID: PMC8977401
- DOI: 10.1101/cshperspect.a040840
Building and Maintaining the Skin
Abstract
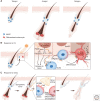
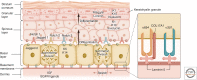
The skin forms a crucial, dynamic barrier between an animal and the external world. In mammals, three stem cell populations possess robust regenerative potential to maintain and repair the body's protective surface: epidermal stem cells, which maintain the stratified epidermis; hair follicle stem cells, which power the cyclic growth of the hair follicle; and melanocyte stem cells, which regenerate pigment-producing melanocytes to color the skin and hair. These stem cells reside in complex microenvironments ("niches") comprising diverse cellular repertoires that enable stem cells to rejuvenate tissues during homeostasis and regenerate them upon injury. Beyond their niches, skin stem cells can also sense and respond to fluctuations in organismal health or changes outside the body. Here, we review these diverse cellular interactions and highlight how far-reaching signals can be transmitted at the local level to enable skin stem cells to tailor their actions to suit the particular occasion and optimize fitness.
Copyright © 2022 Cold Spring Harbor Laboratory Press; all rights reserved.
Figures


References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
