A starting guide to root ecology: strengthening ecological concepts and standardising root classification, sampling, processing and trait measurements
- PMID: 34608637
- PMCID: PMC8518129
- DOI: 10.1111/nph.17572
A starting guide to root ecology: strengthening ecological concepts and standardising root classification, sampling, processing and trait measurements
Erratum in
-
Corrigendum.New Phytol. 2022 Jul;235(1):372. doi: 10.1111/nph.18126. Epub 2022 Apr 28. New Phytol. 2022. PMID: 35478324 Free PMC article. No abstract available.
Abstract
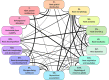
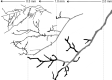
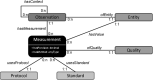
In the context of a recent massive increase in research on plant root functions and their impact on the environment, root ecologists currently face many important challenges to keep on generating cutting-edge, meaningful and integrated knowledge. Consideration of the below-ground components in plant and ecosystem studies has been consistently called for in recent decades, but methodology is disparate and sometimes inappropriate. This handbook, based on the collective effort of a large team of experts, will improve trait comparisons across studies and integration of information across databases by providing standardised methods and controlled vocabularies. It is meant to be used not only as starting point by students and scientists who desire working on below-ground ecosystems, but also by experts for consolidating and broadening their views on multiple aspects of root ecology. Beyond the classical compilation of measurement protocols, we have synthesised recommendations from the literature to provide key background knowledge useful for: (1) defining below-ground plant entities and giving keys for their meaningful dissection, classification and naming beyond the classical fine-root vs coarse-root approach; (2) considering the specificity of root research to produce sound laboratory and field data; (3) describing typical, but overlooked steps for studying roots (e.g. root handling, cleaning and storage); and (4) gathering metadata necessary for the interpretation of results and their reuse. Most importantly, all root traits have been introduced with some degree of ecological context that will be a foundation for understanding their ecological meaning, their typical use and uncertainties, and some methodological and conceptual perspectives for future research. Considering all of this, we urge readers not to solely extract protocol recommendations for trait measurements from this work, but to take a moment to read and reflect on the extensive information contained in this broader guide to root ecology, including sections I-VII and the many introductions to each section and root trait description. Finally, it is critical to understand that a major aim of this guide is to help break down barriers between the many subdisciplines of root ecology and ecophysiology, broaden researchers' views on the multiple aspects of root study and create favourable conditions for the inception of comprehensive experiments on the role of roots in plant and ecosystem functioning.
Keywords: below-ground ecology; handbook; plant root functions; protocol; root classification; root ecology; root traits; trait measurements.
© 2021 The Authors. New Phytologist © 2021 New Phytologist Foundation.
Figures


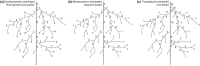

















References
-
- Aarssen LW. 2008. Death without sex—the ‘problem of the small’ and selection for reproductive economy in flowering plants. Evolutionary Ecology 22: 279–298.
-
- Aber JD, Melillo JM, Nadelhoffer KJ, Mcclaugherty CA, Pastor J. 1985. Fine root turnover in forest ecosystems in relation to quantity and form of nitrogen availability – a comparison of two methods. Oecologia 66: 317–321. - PubMed
-
- Abiven S, Heim A, Schmidt MW. 2011. Lignin content and chemical characteristics in maize and wheat vary between plant organs and growth stages: consequences for assessing lignin dynamics in soil. Plant and Soil 343: 369–378.
-
- Abramoff RZ, Finzi AC. 2015. Are above‐and below‐ground phenology in sync? New Phytologist 205: 1054–1061. - PubMed
-
- Abrams MD. 1990. Adaptations and responses to drought in Quercus species of North America. Tree Physiology 7: 227–238. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources

