Cellular junction and mesenchymal factors delineate an endometriosis-specific response of endometrial stromal cells to the mesothelium
- PMID: 34624439
- PMCID: PMC12048087
- DOI: 10.1016/j.mce.2021.111481
Cellular junction and mesenchymal factors delineate an endometriosis-specific response of endometrial stromal cells to the mesothelium
Abstract
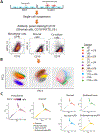
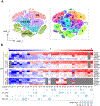
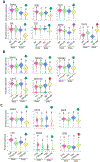
Endometriosis is a debilitating gynecologic disorder that affects ∼10% of women of reproductive age. Endometriosis is characterized by growth of endometriosis lesions within the abdominal cavity, generally thought to arise from retrograde menstruation of shed endometrial tissue. While the pathophysiology underlying peritoneal endometriosis lesion formation is still unclear, the interaction between invading endometrial tissue and the peritoneal mesothelial lining is an essential step in lesion formation. In this study, we assessed proteomic differences between eutopic endometrial stromal cells (ESCs) from women with and without endometriosis in response to peritoneal mesothelial cell (PMC) exposure, using single-cell cytometry by time-of-flight (CyTOF). Co-cultured primary eutopic ESCs from women with and without endometriosis with an established PMC line were subjected to immunostaining with a panel of Maxpar CyTOF metal-conjugated antibodies (n = 28) targeting cell junction and mesenchymal markers, which are involved in cell-cell adhesions and epithelial-mesenchymal transition. Exposure of the ESCs to PMCs resulted in a drastic shift in cellular expression profiles in ESCs derived from endometriosis, whereas little effect by PMCs was observed in ESCs from non-endometriosis subjects. The transcription factor SNAI1 was consistently repressed by PMC interactions. ESCs from endometriosis patients are unique in that they respond to PMCs by undergoing changes in adhesive properties and mesenchymal characteristics that would facilitate lesion formation.
Keywords: Cellular adhesion; CyTOF; Endometriosis; Mass cytometry; Mesenchymal factors.
Copyright © 2021 The Authors. Published by Elsevier B.V. All rights reserved.
Figures


References
-
- Angerer P, Haghverdi L, Buttner M, Theis FJ, Marr C, Buettner F, 2016. destiny: diffusion maps for large-scale single-cell data in R. Bioinformatics 32, 1241–1243. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials

