Efficacy and safety of gemcitabine plus doxorubicin in patients with renal medullary carcinoma
- PMID: 34625389
- PMCID: PMC12289336
- DOI: 10.1016/j.clgc.2021.08.007
Efficacy and safety of gemcitabine plus doxorubicin in patients with renal medullary carcinoma
Abstract
Introduction: Renal medullary carcinoma (RMC) is a rare and lethal renal cell carcinoma characterized by the loss of tumor suppressor SMARCB1. Molecular profiling studies have suggested that RMC cells may be vulnerable to therapies that generate DNA damage, such as the combination of the nucleoside analog gemcitabine, and topoisomerase inhibitor doxorubicin.
Patients and methods: We retrospectively analyzed the records of patients with RMC treated with gemcitabine plus doxorubicin at our institution between January 2005 and September 2020. Best radiographic response and disease progression (RECIST v1.1) were assessed by a blinded radiologist.
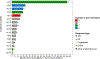
Results: Sixteen patients were included in the study. All but 1 patient (93.8%) received prior platinum-based chemotherapy. Gemcitabine was given intravenously at 900-1200 mg/m2 and doxorubicin at 40-50 mg/m2 intravenously every 2 weeks. Three patients (18.8%) achieved partial response and 7 (43.8%) patients achieved stable disease. The median progression-free survival was 2.8 months (95% CI, 0-6.0). Median overall survival (OS) from gemcitabine plus doxorubicin initiation was 8.1 months (95% CI, 4.6-11.7) and OS from diagnosis was 15.5 months (95% CI, 4.2-26.8 months). There were no grade ≥ 4 AEs; grade 3 AEs were cytopenias (18.8%), nausea (12.5%), fatigue (12.5%), and cardiotoxicity (6.2%). No somatic alterations were detected in the 9 patients tested by targeted next generation sequencing assays.
Conclusion: Gemcitabine plus doxorubicin was well tolerated and demonstrated clinical activity in patients with platinum-refractory RMC, with a subset of patients experiencing durable responses lasting longer than 6 months. Further investigation is warranted to determine biomarkers of sensitivity and target mechanisms of resistance.
Keywords: Gemcitabine plus doxorubicin; Platinum-based chemotherapy; Renal cell carcinoma; Renal medullary carcinoma; SMARCB1.
Copyright © 2021. Published by Elsevier Inc.
Conflict of interest statement
Conflict of Interest Disclosure:
Figures

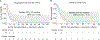
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

