Nestin promotes pulmonary fibrosis via facilitating recycling of TGF-β receptor I
- PMID: 34625478
- PMCID: PMC9068978
- DOI: 10.1183/13993003.03721-2020
Nestin promotes pulmonary fibrosis via facilitating recycling of TGF-β receptor I
Abstract
Background: Idiopathic pulmonary fibrosis (IPF) is a progressive fibrotic lung disease that is characterised by aberrant proliferation of activated myofibroblasts and pathological remodelling of the extracellular matrix. Previous studies have revealed that the intermediate filament protein nestin plays key roles in tissue regeneration and wound healing in different organs. Whether nestin plays a critical role in the pathogenesis of IPF needs to be clarified.
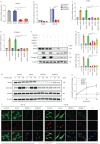
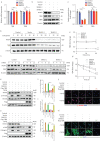
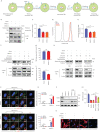
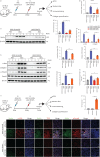
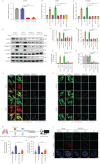
Methods: Nestin expression in lung tissues from bleomycin-treated mice and IPF patients was determined. Transfection with nestin short hairpin RNA vectors in vitro that regulated transcription growth factor (TGF)-β/Smad signalling was conducted. Biotinylation assays to observe plasma membrane TβRI, TβRI endocytosis and TβRI recycling after nestin knockdown were performed. Adeno-associated virus serotype (AAV)6-mediated nestin knockdown was assessed in vivo.
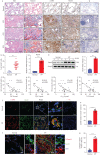
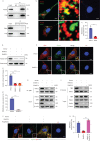
Results: We found that nestin expression was increased in a murine pulmonary fibrosis model and IPF patients, and that the upregulated protein primarily localised in lung α-smooth muscle actin-positive myofibroblasts. Mechanistically, we determined that nestin knockdown inhibited TGF-β signalling by suppressing recycling of TβRI to the cell surface and that Rab11 was required for the ability of nestin to promote TβRI recycling. In vivo, we found that intratracheal administration of AAV6-mediated nestin knockdown significantly alleviated pulmonary fibrosis in multiple experimental mice models.
Conclusion: Our findings reveal a pro-fibrotic function of nestin partially through facilitating Rab11-dependent recycling of TβRI and shed new light on pulmonary fibrosis treatment.
Copyright ©The authors 2022.
Conflict of interest statement
Conflict of interest: J. Wang has nothing to disclose. Conflict of interest: X. Lai has nothing to disclose. Conflict of interest: S. Yao has nothing to disclose. Conflict of interest: H. Chen has nothing to disclose. Conflict of interest: J. Cai has nothing to disclose. Conflict of interest: Y. Luo has nothing to disclose. Conflict of interest: Y. Wang has nothing to disclose. Conflict of interest: Y. Qiu has nothing to disclose. Conflict of interest: Y. Huang has nothing to disclose. Conflict of interest: X. Wei has nothing to disclose. Conflict of interest: B. Wang has nothing to disclose. Conflict of interest: Q. Lu has nothing to disclose. Conflict of interest: Y. Guan has nothing to disclose. Conflict of interest: T. Wang has nothing to disclose. Conflict of interest: S. Li has nothing to disclose. Conflict of interest: A.P. Xiang has nothing to disclose.
Figures

Comment in
-
Targeting the nasty nestin to shoot lung fibrosis.Eur Respir J. 2022 May 5;59(5):2103146. doi: 10.1183/13993003.03146-2021. Print 2022 May. Eur Respir J. 2022. PMID: 35512809 No abstract available.
References
-
- Travis WD, Costabel U, Hansell DM, et al. . An official American Thoracic Society/European Respiratory Society statement: update of the international multidisciplinary classification of the idiopathic interstitial pneumonias. Am J Respir Crit Care Med 2013; 188: 733–748. doi:10.1164/rccm.201308-1483ST - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
