Extended-release calcifediol in stage 3-4 chronic kidney disease: a new therapy for the treatment of secondary hyperparathyroidism associated with hypovitaminosis D
- PMID: 34626363
- PMCID: PMC8995284
- DOI: 10.1007/s40620-021-01152-5
Extended-release calcifediol in stage 3-4 chronic kidney disease: a new therapy for the treatment of secondary hyperparathyroidism associated with hypovitaminosis D
Abstract
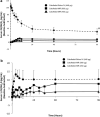
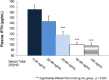
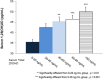
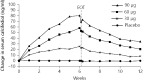
A high percentage of patients with chronic kidney disease have hypovitaminosis D, which is a driver of secondary hyperparathyroidism and an important factor in chronic kidney disease-mineral and bone disorder. Vitamin D deficiency (serum total 25-OH vitamin D levels < 30 ng/mL) occurs early in the course of chronic kidney disease and treatment guidelines recommend early intervention to restore 25-OH vitamin D levels as a first step to prevent/delay the onset/progression of secondary hyperparathyroidism. The vitamin D forms administered to replace 25-OH vitamin D include cholecalciferol, ergocalciferol, and immediate- or extended-release formulations of calcifediol. Most patients with intermediate-stage chronic kidney disease will develop secondary hyperparathyroidism before dialysis is required. Control of parathyroid hormone levels becomes a major focus of therapy in these patients. This article focuses on the position of extended-release calcifediol in the treatment of patients with stage 3-4 chronic kidney disease and secondary hyperparathyroidism with hypovitaminosis D. Several characteristics of extended-release calcifediol support its use in the intermediate stages of chronic kidney disease. The pharmacokinetics of extended-release calcifediol make it effective for replenishing 25-OH vitamin D levels, with minimal impact on vitamin D catabolism from fibroblast-growth factor-23 and CYP24A1 upregulation. Extended-release calcifediol increases circulating 25-OH vitamin D levels in a dose-dependent manner and lowers parathyroid hormone levels by a clinically relevant extent, comparable to what can be achieved by administering active vitamin D analogues, though with a lower risk of hypercalcaemia and hyperphosphataemia. Active vitamin D analogues are reserved for patients undergoing dialysis or pre-dialysis patients with severe progressive secondary hyperparathyroidism.
Keywords: Calcifediol; Chronic kidney disease; Parathyroid hormone; Secondary hyperparathyroidism; Vitamin D; Vitamin D insufficiency.
© 2021. The Author(s).
Conflict of interest statement
MC declares advisory/lecture fees from Amgen, Abbvie, Shire, Vifor-Pharma, and Baxter. PM declares no competing interest. PN has attended a board meeting supported by Vifor Pharma.
Figures
Similar articles
-
Extended-Release Calcifediol Effectively Raises Serum Total 25-Hydroxyvitamin D Even in Overweight Nondialysis Chronic Kidney Disease Patients with Secondary Hyperparathyroidism.Am J Nephrol. 2022;53(6):446-454. doi: 10.1159/000524289. Epub 2022 May 12. Am J Nephrol. 2022. PMID: 35551374 Free PMC article.
-
Calcifediol to treat secondary hyperparathyroidism in patients with chronic kidney disease.Expert Rev Clin Pharmacol. 2017 Oct;10(10):1073-1084. doi: 10.1080/17512433.2017.1371011. Epub 2017 Sep 4. Expert Rev Clin Pharmacol. 2017. PMID: 28846459 Review.
-
Extended-Release Calcifediol: A Data Journey from Phase 3 Studies to Real-World Evidence Highlights the Importance of Early Treatment of Secondary Hyperparathyroidism.Nephron. 2024;148(10):657-666. doi: 10.1159/000538818. Epub 2024 Apr 24. Nephron. 2024. PMID: 38657576 Free PMC article. Review.
-
Correction of 25-OH-vitamin D deficiency improves control of secondary hyperparathyroidism and reduces the inflammation in stable haemodialysis patients.Nefrologia (Engl Ed). 2018 Jan-Feb;38(1):41-47. doi: 10.1016/j.nefro.2017.05.008. Epub 2017 Jul 1. Nefrologia (Engl Ed). 2018. PMID: 28673686 English, Spanish.
-
Rationale for Raising Current Clinical Practice Guideline Target for Serum 25-Hydroxyvitamin D in Chronic Kidney Disease.Am J Nephrol. 2019;49(4):284-293. doi: 10.1159/000499187. Epub 2019 Mar 15. Am J Nephrol. 2019. PMID: 30878999 Clinical Trial.
Cited by
-
Calcifediol in the management of vitamin D deficiency-related skeletal and extraskeletal diseases: overview and clinical cases.Drugs Context. 2023 Sep 6;12:2023-5-4. doi: 10.7573/dic.2023-5-4. eCollection 2023. Drugs Context. 2023. PMID: 37711731 Free PMC article. Review.
-
Calcifediol and paricalcitol as adjunctive therapies for HSV-1 keratitis and corneal perforation: A case report.Medicine (Baltimore). 2024 Dec 6;103(49):e40654. doi: 10.1097/MD.0000000000040654. Medicine (Baltimore). 2024. PMID: 39654170 Free PMC article.
-
Recurrence hyperparathyroidism caused by synchronous parathyroid carcinoma and parathyromatosis in a patient with long-term hemodialysis.BMC Nephrol. 2023 Oct 4;24(1):293. doi: 10.1186/s12882-023-03328-6. BMC Nephrol. 2023. PMID: 37794407 Free PMC article.
-
Role of vitamin D supplementation in the management of musculoskeletal diseases: update from an European Society of Clinical and Economical Aspects of Osteoporosis, Osteoarthritis and Musculoskeletal Diseases (ESCEO) working group.Aging Clin Exp Res. 2022 Nov;34(11):2603-2623. doi: 10.1007/s40520-022-02279-6. Epub 2022 Oct 26. Aging Clin Exp Res. 2022. PMID: 36287325 Free PMC article. Review.
-
Vitamin D: are all compounds equal?Clin Kidney J. 2025 Mar 13;18(Suppl 1):i61-i96. doi: 10.1093/ckj/sfae417. eCollection 2025 Mar. Clin Kidney J. 2025. PMID: 40083955 Free PMC article.
References
-
- Ketteler M, Block GA, Evenepoel P, et al. Diagnosis, evaluation, prevention, and treatment of chronic kidney disease-mineral and bone disorder: synopsis of the kidney disease: improving global outcomes 2017 clinical practice guideline update. Ann Intern Med. 2018;168:422–430. doi: 10.7326/M17-2640. - DOI - PubMed

