A Zebrafish Model of Metastatic Colonization Pinpoints Cellular Mechanisms of Circulating Tumor Cell Extravasation
- PMID: 34631514
- PMCID: PMC8495265
- DOI: 10.3389/fonc.2021.641187
A Zebrafish Model of Metastatic Colonization Pinpoints Cellular Mechanisms of Circulating Tumor Cell Extravasation
Abstract
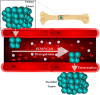
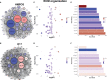
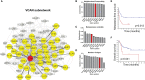
Metastasis is a multistep process in which cells must detach, migrate/invade local structures, intravasate, circulate, extravasate, and colonize. A full understanding of the complexity of this process has been limited by the lack of ability to study these steps in isolation with detailed molecular analyses. Leveraging a comparative oncology approach, we injected canine osteosarcoma cells into the circulation of transgenic zebrafish with fluorescent blood vessels in a biologically dynamic metastasis extravasation model. Circulating tumor cell clusters that successfully extravasated the vasculature as multicellular units were isolated under intravital imaging (n = 6). These extravasation-positive tumor cell clusters sublines were then molecularly profiled by RNA-Seq. Using a systems-level analysis, we pinpointed the downregulation of KRAS signaling, immune pathways, and extracellular matrix (ECM) organization as enriched in extravasated cells (p < 0.05). Within the extracellular matrix remodeling pathway, we identified versican (VCAN) as consistently upregulated and central to the ECM gene regulatory network (p < 0.05). Versican expression is prognostic for a poorer metastasis-free and overall survival in patients with osteosarcoma. Together, our results provide a novel experimental framework to study discrete steps in the metastatic process. Using this system, we identify the versican/ECM network dysregulation as a potential contributor to osteosarcoma circulating tumor cell metastasis.
Keywords: angiopellosis; cancer exodus hypothesis; circulating tumor cell cluster; metastasis; osteosarcoma; tumor cell extravasation.
Copyright © 2021 Allen, Cullen, Hawkey, Mochizuki, Nguyen, Schechter, Borst, Yoder, Freedman, Patierno, Cheng, Eward and Somarelli.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


References
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

