Non-polar lipid from greenshell mussel (Perna canaliculus) inhibits osteoclast differentiation
- PMID: 34632003
- PMCID: PMC8493498
- DOI: 10.1016/j.bonr.2021.101132
Non-polar lipid from greenshell mussel (Perna canaliculus) inhibits osteoclast differentiation
Abstract
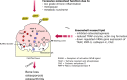
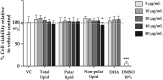
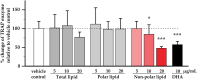
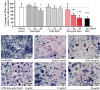
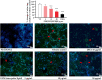
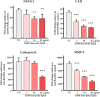
The osteoclast-dependent bone resorption process is a crucial part of the bone regulatory system. The excessive function of osteoclasts can cause diseases of bone, joint, and other tissues such as osteoporosis and osteoarthritis. Greenshell mussel oil (GSM), a good source of long chain omega-3 polyunsaturated fatty acids (LCn-3PUFAs), was fractionated into total lipid, polar lipid, and non-polar lipid components and their anti-osteoclastogenic activity tested in RAW 264.7 cell cultures. Osteoclast differentiation process was achieved after 5 days of incubation with RANKL in 24-well culture plates. Introducing the non-polar lipid fraction into the culture caused a lack of cell differentiation, and a reduction in tartrate-resistant acid phosphatase (TRAP) activity and TRAP cell numbers in a dose-dependent manner (50% reduction at the concentration of 20 μg/mL, p < 0.001). Moreover, actin ring formation was significantly diminished by non-polar lipids at 10-20 μg/mL. The bone digestive enzymes released by osteoclasts into the pit formation were also compromised by downregulating gene expression of cathepsin K, carbonic anhydrase II (CA II), matrix metalloproteinase 9 (MMP-9), and nuclear factor of activated T-cells, cytoplasmic 1 (NFATc1). This study revealed that the non-polar lipid fraction of GSM oil contains bioactive substances which possess potent anti-osteoclastogenic activity.
Keywords: AA, Arachidonic acid; ALA, Alpha linolenic acid; CAII, Carbonic anhydrase II; DHA, Docosahexaenoic acid; DMSO, dimethyl sulfoxide; DPA, Docosapentaenoic acid; EPA, Eicosapentaenoic acid; FFAR, Free fatty acid receptor; GSM, Greenshell mussel; Greenshell mussel; LA, Linoleic acid; LPS, Lipopolysaccharide; MMP-9, Matrix metalloproteinase 9; MUFA, Monounsaturated fatty acid; NF-κB, Nuclear factor κB; NFATc1, Nuclear factor of activated T-cells, cytoplasmic 1; OA, Osteoarthritis; Omega 3 fatty acid; Osteoarthritis; Osteoclasts; Osteoporosis; PA, Palmitic acid; PPAR, Peroxisome proliferator activated receptor; PUFA, Polyunsaturated fatty acid; RANKL, Receptor activator of nuclear factor κB ligand; SFA, Saturated fatty acid; TRAP, Tartrate-resistant acid phosphatase.
© 2021 Published by Elsevier Inc.
Conflict of interest statement
H.S.T. has a conflict of interest as she is employed by Sanford Limited which is a mussel producer and exporter.
Figures
Similar articles
-
Docosahexaenoic acid is more potent inhibitor of osteoclast differentiation in RAW 264.7 cells than eicosapentaenoic acid.J Cell Physiol. 2008 Jan;214(1):201-9. doi: 10.1002/jcp.21188. J Cell Physiol. 2008. PMID: 17929247
-
Effects of ω3- and ω6-polyunsaturated fatty acids on RANKL-induced osteoclast differentiation of RAW264.7 cells: a comparative in vitro study.Nutrients. 2014 Jul 9;6(7):2584-601. doi: 10.3390/nu6072584. Nutrients. 2014. PMID: 25010555 Free PMC article.
-
Receptor activator of NF-kappaB ligand induces the expression of carbonic anhydrase II, cathepsin K, and matrix metalloproteinase-9 in osteoclast precursor RAW264.7 cells.Life Sci. 2007 Mar 13;80(14):1311-8. doi: 10.1016/j.lfs.2006.12.037. Epub 2007 Jan 23. Life Sci. 2007. PMID: 17306833
-
n-3 Polyunsaturated fatty acids for the management of alcoholic liver disease: A critical review.Crit Rev Food Sci Nutr. 2019;59(sup1):S116-S129. doi: 10.1080/10408398.2018.1544542. Epub 2018 Dec 22. Crit Rev Food Sci Nutr. 2019. PMID: 30580553 Review.
-
Potential modulatory mechanisms of action by long-chain polyunsaturated fatty acids on bone cell and chondrocyte metabolism.Prog Lipid Res. 2021 Jul;83:101113. doi: 10.1016/j.plipres.2021.101113. Epub 2021 Jul 2. Prog Lipid Res. 2021. PMID: 34217732 Review.
Cited by
-
The Influence of Obesity, Ovariectomy, and Greenshell Mussel Supplementation on Bone Mineral Density in Rats.JBMR Plus. 2021 Nov 14;6(1):e10571. doi: 10.1002/jbm4.10571. eCollection 2022 Jan. JBMR Plus. 2021. PMID: 35079679 Free PMC article.
-
Effects and Mechanisms of Polyunsaturated Fatty Acids on Age-Related Musculoskeletal Diseases: Sarcopenia, Osteoporosis, and Osteoarthritis-A Narrative Review.Nutrients. 2024 Sep 16;16(18):3130. doi: 10.3390/nu16183130. Nutrients. 2024. PMID: 39339730 Free PMC article. Review.
References
-
- Bertuglia A., Lacourt M., Girard C., Beauchamp G., Richard H., Laverty S. Osteoclasts are recruited to the subchondral bone in naturally occurring post-traumatic equine carpal osteoarthritis and may contribute to cartilage degradation. Osteoarthr. Cartil. 2016;24(3):555–566. doi: 10.1016/j.joca.2015.10.008. - DOI - PubMed
-
- Boeyens J.C., Deepak V., Chua W.H., Kruger M.C., Joubert A.M., Coetzee M. Effects of omega3- and omega6-polyunsaturated fatty acids on RANKL-induced osteoclast differentiation of RAW264.7 cells: a comparative in vitro study. Nutrients. 2014;6(7):2584–2601. doi: 10.3390/nu6072584. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

