Evaluation of [18F]PI-2620, a second-generation selective tau tracer, for assessing four-repeat tauopathies
- PMID: 34632382
- PMCID: PMC8495135
- DOI: 10.1093/braincomms/fcab190
Evaluation of [18F]PI-2620, a second-generation selective tau tracer, for assessing four-repeat tauopathies
Abstract
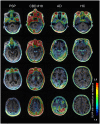
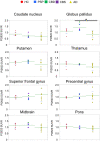
Tau aggregates represent a key pathologic feature of Alzheimer's disease and other neurodegenerative diseases. Recently, PET probes have been developed for in vivo detection of tau accumulation; however, they are limited because of off-target binding and a reduced ability to detect tau in non-Alzheimer's disease tauopathies. The novel tau PET tracer, [18F]PI-2620, has a high binding affinity and specificity for aggregated tau; therefore, it was hypothesized to have desirable properties for the visualization of tau accumulation in Alzheimer's disease and non-Alzheimer's disease tauopathies. To assess the ability of [18F]PI-2620 to detect regional tau burden in non-Alzheimer's disease tauopathies compared with Alzheimer's disease, patients with progressive supranuclear palsy (n = 3), corticobasal syndrome (n = 2), corticobasal degeneration (n = 1) or Alzheimer's disease (n = 8), and healthy controls (n = 7) were recruited. All participants underwent MRI, amyloid β assessment and [18F]PI-2620 PET (Image acquisition at 60-90 min post-injection). Cortical and subcortical tau accumulations were assessed by calculating standardized uptake value ratios using [18F]PI-2620 PET. For pathologic validation, tau pathology was assessed using tau immunohistochemistry and compared with [18F]PI-2620 retention in an autopsied case of corticobasal degeneration. In Alzheimer's disease, focal retention of [18F]PI-2620 was evident in the temporal and parietal lobes, precuneus, and cingulate cortex. Standardized uptake value ratio analyses revealed that patients with non-Alzheimer's disease tauopathies had elevated [18F]PI-2620 uptake only in the globus pallidus, as compared to patients with Alzheimer's disease, but not healthy controls. A head-to-head comparison of [18F]PI-2620 and [18F]PM-PBB3, another tau PET probe for possibly visualizing the four-repeat tau pathogenesis in non-Alzheimer's disease, revealed different retention patterns in one subject with progressive supranuclear palsy. Imaging-pathology correlation analysis of the autopsied patient with corticobasal degeneration revealed no significant correlation between [18F]PI-2620 retention in vivo. High [18F]PI-2620 uptake at 60-90 min post-injection in the globus pallidus may be a sign of neurodegeneration in four-repeat tauopathy, but not necessarily practical for diagnosis of non-Alzheimer's disease tauopathies. Collectively, this tracer is a promising tool to detect Alzheimer's disease-tau aggregation. However, late acquisition PET images of [18F]PI-2620 may have limited utility for reliable detection of four-repeat tauopathy because of lack of correlation between post-mortem tau pathology and different retention pattern than the non-Alzheimer's disease-detectable tau radiotracer, [18F]PM-PBB3. A recent study reported that [18F]PI-2620 tracer kinetics curves in four-repeat tauopathies peak earlier (within 30 min) than Alzheimer's disease; therefore, further studies are needed to determine appropriate PET acquisition times that depend on the respective interest regions and diseases.
Keywords: Alzheimer’s disease; Tau imaging; corticobasal degeneration; progressive supranuclear palsy; tauopathy.
© The Author(s) (2021). Published by Oxford University Press on behalf of the Guarantors of Brain.
Figures



Similar articles
-
Symptomatology in 4-repeat tauopathies is associated with data-driven topology of [18F]-PI-2620 tau-PET signal.Neuroimage Clin. 2023;38:103402. doi: 10.1016/j.nicl.2023.103402. Epub 2023 Apr 11. Neuroimage Clin. 2023. PMID: 37087820 Free PMC article.
-
18F-flortaucipir PET to autopsy comparisons in Alzheimer's disease and other neurodegenerative diseases.Brain. 2020 Dec 5;143(11):3477-3494. doi: 10.1093/brain/awaa276. Brain. 2020. PMID: 33141172 Free PMC article.
-
Subcortical tau is linked to hypoperfusion in connected cortical regions in 4-repeat tauopathies.Brain. 2024 Jul 5;147(7):2428-2439. doi: 10.1093/brain/awae174. Brain. 2024. PMID: 38842726
-
Current Progress and Future Directions in Non-Alzheimer's Disease Tau PET Tracers.ACS Chem Neurosci. 2025 Jan 15;16(2):111-127. doi: 10.1021/acschemneuro.4c00319. Epub 2025 Jan 6. ACS Chem Neurosci. 2025. PMID: 39762194 Review.
-
Imaging tau pathology in Parkinsonisms.NPJ Parkinsons Dis. 2017 Jun 29;3:22. doi: 10.1038/s41531-017-0023-3. eCollection 2017. NPJ Parkinsons Dis. 2017. PMID: 28685158 Free PMC article. Review.
Cited by
-
Ligands for Protein Fibrils of Amyloid-β, α-Synuclein, and Tau.Chem Rev. 2025 Jun 11;125(11):5282-5348. doi: 10.1021/acs.chemrev.4c00838. Epub 2025 May 6. Chem Rev. 2025. PMID: 40327808 Free PMC article. Review.
-
Amyloid and Tau Positron Emission Tomography Imaging in Alzheimer's Disease and Other Tauopathies.Front Aging Neurosci. 2022 Apr 22;14:838034. doi: 10.3389/fnagi.2022.838034. eCollection 2022. Front Aging Neurosci. 2022. PMID: 35527737 Free PMC article. Review.
-
In vivo PET classification of tau pathologies in patients with frontotemporal dementia.Brain Commun. 2024 Mar 1;6(2):fcae075. doi: 10.1093/braincomms/fcae075. eCollection 2024. Brain Commun. 2024. PMID: 38510212 Free PMC article.
-
Radiation dosimetry and pharmacokinetics of the tau PET tracer florzolotau (18F) in healthy Japanese subjects.Ann Nucl Med. 2023 May;37(5):300-309. doi: 10.1007/s12149-023-01828-x. Epub 2023 Mar 9. Ann Nucl Med. 2023. PMID: 36890399 Free PMC article.
-
Staging of progressive supranuclear palsy-Richardson syndrome using MRI brain charts for the human lifespan.Brain Commun. 2024 Feb 20;6(2):fcae055. doi: 10.1093/braincomms/fcae055. eCollection 2024. Brain Commun. 2024. PMID: 38444913 Free PMC article.
References
-
- Klunk WE, Engler H, Nordberg A, et al.Imaging brain amyloid in Alzheimer's disease with Pittsburgh Compound-B. Ann Neurol. 2004;55(3):306–319. - PubMed
-
- Lohith TG, Bennacef I, Vandenberghe R, et al.Brain imaging of Alzheimer dementia patients and elderly controls with (18)F-MK-6240, a PET tracer targeting neurofibrillary tangles. J Nucl Med. 2019;60(1):107–114. - PubMed
-
- Tagai K, Ono M, Kubota M, et al.High-contrast in vivo imaging of tau pathologies in Alzheimer's and non-Alzheimer's disease tauopathies. Neuron. 2021;109(1):42–58 e8. - PubMed
-
- Takahata K, Kimura Y, Sahara N, et al.PET-detectable tau pathology correlates with long-term neuropsychiatric outcomes in patients with traumatic brain injury. Brain. 2019;142(10):3265–3279. - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
