Larval ecology and bionomics of Anopheles funestus in highland and lowland sites in western Kenya
- PMID: 34634069
- PMCID: PMC8504749
- DOI: 10.1371/journal.pone.0255321
Larval ecology and bionomics of Anopheles funestus in highland and lowland sites in western Kenya
Abstract
Background: An. funestus is a major Afrotropical vector of human malaria. This study sought to investigate the larval ecology, sporozoite infection rates and blood meal sources of An. funestus in western Kenya.
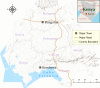
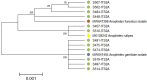
Methods: Larval surveys were carried out in Bungoma (Highland) and Kombewa (lowland) of western Kenya. Aquatic habitats were identified, characterized, georeferenced and carefully examined for mosquito larvae and predators. Indoor resting mosquitoes were sampled using pyrethrum spray catches. Adults and larvae were morphologically and molecularly identified to species. Sporozoite infections and blood meal sources were detected using real-time PCR and ELISA respectively.
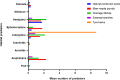
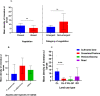
Results: Of the 151 aquatic habitats assessed, 62/80 (78%) in Bungoma and 58/71(82%) in Kombewa were positive for mosquito larvae. Of the 3,193 larvae sampled, An. funestus larvae constitute 38% (1224/3193). Bungoma recorded a higher number of An. funestus larvae (85%, 95%, CI, 8.722-17.15) than Kombewa (15%, 95%, CI, 1.33-3.91). Molecular identification of larvae showed that 89% (n = 80) were An. funestus. Approximately 59%, 35% and 5% of An. funestus larvae co-existed with An. gambiae s.l, Culex spp and An. coustani in the same habitats respectively. Of 1,221 An. funestus s.l adults sampled, molecular identifications revealed that An. funestus constituted 87% (n = 201) and 88% (n = 179) in Bungoma and Kombewa, respectively. The Plasmodium falciparum sporozoite rate of An. funestus in Bungoma and Kombewa was 2% (3/174) and 1% (2/157), respectively, and the human blood index of An. funestus was 84% (48/57) and 89% (39/44) and for Bungoma and Kombewa, respectively.
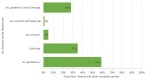
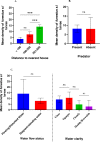
Conclusion: Man-made ponds had the highest abundance of An. funestus larvae. Multiple regression and principal component analyses identified the distance to the nearest house as the key environmental factor associated with the abundance of An. funestus larvae in aquatic habitats. This study serves as a guide for the control of An. funestus and other mosquito species to complement existing vector control strategies.
Conflict of interest statement
The authors have no conflicts of interest to declare.
Figures
References
-
- WHO, World malaria report 2020: 20 years of global progress and challenges. Geneva: World Health Organization; 2020. Licence: CC BY-NC-SA 3.0 IGO. 2020.
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

