CAM photosynthesis: the acid test
- PMID: 34637529
- PMCID: PMC9298356
- DOI: 10.1111/nph.17790
CAM photosynthesis: the acid test
Abstract
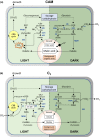
There is currently considerable interest in the prospects for bioengineering crassulacean acid metabolism (CAM) photosynthesis - or key elements associated with it, such as increased water-use efficiency - into C3 plants. Resolving how CAM photosynthesis evolved from the ancestral C3 pathway could provide valuable insights into the targets for such bioengineering efforts. It has been proposed that the ability to accumulate organic acids at night may be common among C3 plants, and that the transition to CAM might simply require enhancement of pre-existing fluxes, without the need for changes in circadian or diurnal regulation. We show, in a survey encompassing 40 families of vascular plants, that nocturnal acidification is a feature entirely restricted to CAM species. Although many C3 species can synthesize malate during the light period, we argue that the switch to night-time malic acid accumulation requires a fundamental metabolic reprogramming that couples glycolytic breakdown of storage carbohydrate to the process of net dark CO2 fixation. This central element of the CAM pathway, even when expressed at a low level, represents a biochemical capability not seen in C3 plants, and so is better regarded as a discrete evolutionary innovation than as part of a metabolic continuum between C3 and CAM.
Keywords: C3 photosynthesis; CAM photosynthesis; carboxylate; citrate; crassulacean acid metabolism; malate; malic acid; titratable acidity.
© 2021 The Authors. New Phytologist © 2021 New Phytologist Foundation This article has been contributed to by US Government employees and their work is in the public domain in the USA.
Figures
References
-
- Abraham PE, Yin H, Borland AM, Weighill D, Lim SD, De Paoli HC, Engle N, Jones PC, Agh R, Weston DJ et al. 2016. Transcript, protein and metabolite temporal dynamics in the CAM plant Agave . Nature Plants 2: 16178. - PubMed
-
- Amagasa T. 1982. The influence of leaf age on the diurnal changes of malate and starch in the CAM plant Kalanchoe daigremontiana Hamet et Perr. Zeitschrift für Pflanzenphysiologie 108: 93–96.
-
- Andrés Z, Pérez‐Hormaeche J, Leidi EO, Schlücking K, Steinhorst L, McLachlan DH, Schumacher K, Hetherington AM, Kudla J, Cubero B et al. 2014. Control of vacuolar dynamics and regulation of stomatal aperture by tonoplast potassium uptake. Proceedings of the National Academy of Sciences, USA 111: E1806–E1814. - PMC - PubMed
-
- APG IV . 2016. An update of the Angiosperm Phylogeny Group classification for the orders and families of flowering plants: APG IV. Botanical Journal of the Linnean Society 181: 1–20.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

