Defining the Role of GLI/Hedgehog Signaling in Chemoresistance: Implications in Therapeutic Approaches
- PMID: 34638233
- PMCID: PMC8507559
- DOI: 10.3390/cancers13194746
Defining the Role of GLI/Hedgehog Signaling in Chemoresistance: Implications in Therapeutic Approaches
Abstract
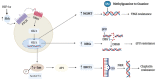
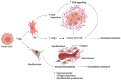
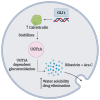
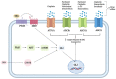
Insight into cancer signaling pathways is vital in the development of new cancer treatments to improve treatment efficacy. A relatively new but essential developmental signaling pathway, namely Hedgehog (Hh), has recently emerged as a major mediator of cancer progression and chemoresistance. The evolutionary conserved Hh signaling pathway requires an in-depth understanding of the paradigm of Hh signaling transduction, which is fundamental to provide the necessary means for the design of novel tools for treating cancer related to aberrant Hh signaling. This review will focus substantially on the canonical Hh signaling and the treatment strategies employed in different studies, with special emphasis on the molecular mechanisms and combination treatment in regard to Hh inhibitors and chemotherapeutics. We discuss our views based on Hh signaling's role in regulating DNA repair machinery, autophagy, tumor microenvironment, drug inactivation, transporters, epithelial-to-mesenchymal transition, and cancer stem cells to promote chemoresistance. The understanding of this Achilles' Heel in cancer may improve the therapeutic outcome for cancer therapy.
Keywords: cancer; chemoresistance; hedgehog inhibitors; hedgehog pathway; molecular mechanism.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

