Hyaluronan and Derivatives: An In Vitro Multilevel Assessment of Their Potential in Viscosupplementation
- PMID: 34641024
- PMCID: PMC8512809
- DOI: 10.3390/polym13193208
Hyaluronan and Derivatives: An In Vitro Multilevel Assessment of Their Potential in Viscosupplementation
Abstract
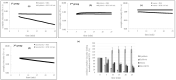
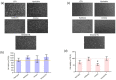
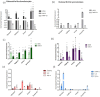
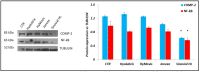
In this research work, viscosupplements based on linear, derivatized, crosslinked and complexed HA forms were extensively examined, providing data on the hydrodynamic parameters for the water-soluble-HA-fraction, rheology, sensitivity to enzymatic hydrolysis and capacity to modulate specific biomarkers' expression in human pathological chondrocytes and synoviocytes. Soluble HA ranged from 0 to 32 mg/mL and from 150 to 1330 kDa MW. The rheological behavior spanned from purely elastic to viscoelastic, suggesting the diversity of the categories that are suitable for restoring specific/different features of the healthy synovial fluid. The rheological parameters were reduced in a diverse manner upon dilution and hyaluronidases action, indicating different durations of the viscosupplementation effect. Bioactivity was found for all the samples, increasing the expression of different matrix markers (e.g., hyaluronan-synthase); however, the hybrid cooperative complexes performed better in most of the experiments. Hybrid cooperative complexes improved COLII mRNA expression (~12-fold increase vs. CTR), proved the most effective at preserving cell phenotype. In addition, in these models, the HA samples reduced inflammation. IL-6 was down-regulated vs. CTR by linear and chemically modified HA, and especially by hybrid complexes. The results represent the first comprehensive panel of data directly comparing the diverse HA forms for intra-articular injections and provide valuable information for tailoring products' clinical use as well as for designing new, highly performing HA-formulations that can address specific needs.
Keywords: OA biomarkers; human chondrocytes; human sinoviocytes; hyaluronan; hyaluronidase; intra-articular injection; rheology.
Conflict of interest statement
CS and ALG are among the inventors, but they are not assignees, of the patent number WO Patent WO/2012/032,151 that describes the novel process preparations undertaken to obtain the HA complexes. Schiraldi’s group has been involved in projects funded by MIUR and MISE with such industrial partners as Altergon Italia s.r.l. and IBSA Italia. All other authors declare no competing interests.
Figures


Similar articles
-
Platelet Rich Plasma and Hyaluronic Acid Blend for the Treatment of Osteoarthritis: Rheological and Biological Evaluation.PLoS One. 2016 Jun 16;11(6):e0157048. doi: 10.1371/journal.pone.0157048. eCollection 2016. PLoS One. 2016. PMID: 27310019 Free PMC article.
-
Potential mechanism of action of intra-articular hyaluronan therapy in osteoarthritis: are the effects molecular weight dependent?Semin Arthritis Rheum. 2002 Aug;32(1):10-37. doi: 10.1053/sarh.2002.33720. Semin Arthritis Rheum. 2002. PMID: 12219318 Review.
-
Hyaluronan viscosupplementation: state of the art and insight into the novel cooperative hybrid complexes based on high and low molecular weight HA of potential interest in osteoarthritis treatment.Clin Cases Miner Bone Metab. 2016 Jan-Apr;13(1):36-7. doi: 10.11138/ccmbm/2016.13.1.036. Epub 2016 May 11. Clin Cases Miner Bone Metab. 2016. PMID: 27252742 Free PMC article. Review.
-
Non-clinical assessment of lubrication and free radical scavenging of an innovative non-animal carboxymethyl chitosan biomaterial for viscosupplementation: An in-vitro and ex-vivo study.PLoS One. 2021 Oct 11;16(10):e0256770. doi: 10.1371/journal.pone.0256770. eCollection 2021. PLoS One. 2021. PMID: 34634053 Free PMC article.
-
A Comparison Between Rheological Properties of Intra-articular Hyaluronic Acid Preparations and Reported Human Synovial Fluid.Adv Ther. 2018 Apr;35(4):523-530. doi: 10.1007/s12325-018-0688-y. Epub 2018 Mar 14. Adv Ther. 2018. PMID: 29542009 Free PMC article.
Cited by
-
Chemical Modification of Hyaluronan and Their Biomedical Applications.Front Chem. 2022 Feb 11;10:830671. doi: 10.3389/fchem.2022.830671. eCollection 2022. Front Chem. 2022. PMID: 35223772 Free PMC article. Review.
-
Potential of Biofermentative Unsulfated Chondroitin and Hyaluronic Acid in Dermal Repair.Int J Mol Sci. 2022 Feb 1;23(3):1686. doi: 10.3390/ijms23031686. Int J Mol Sci. 2022. PMID: 35163608 Free PMC article.
-
Molecular Fingerprint of Human Pathological Synoviocytes in Response to Extractive Sulfated and Biofermentative Unsulfated Chondroitins.Int J Mol Sci. 2022 Dec 14;23(24):15865. doi: 10.3390/ijms232415865. Int J Mol Sci. 2022. PMID: 36555507 Free PMC article.
-
Intra-Articular Hyaluronic Acid for Knee Osteoarthritis: A Systematic Umbrella Review.J Clin Med. 2025 Feb 14;14(4):1272. doi: 10.3390/jcm14041272. J Clin Med. 2025. PMID: 40004802 Free PMC article. Review.
-
Alterations in the Structure, Composition, and Organization of Galactosaminoglycan-Containing Proteoglycans and Collagen Correspond to the Progressive Stages of Dupuytren's Disease.Int J Mol Sci. 2024 Jun 29;25(13):7192. doi: 10.3390/ijms25137192. Int J Mol Sci. 2024. PMID: 39000302 Free PMC article.
References
-
- Fam H., Bryant J.T., Kontopoulou M. Rheological properties of synovial fluids. Biorheology. 2007;44:59–74. - PubMed
-
- Bhuanantanondh P., Grecov D., Kwok E., Guy P. Rheology of osteoarthritic synovial fluid mixed with viscosupplements: A pilot study. Biomed. Eng. Lett. 2011;1:213–219. doi: 10.1007/s13534-011-0034-7. - DOI
LinkOut - more resources
Full Text Sources

