Semisynthetic Derivatives of Selected Amaryllidaceae Alkaloids as a New Class of Antimycobacterial Agents
- PMID: 34641567
- PMCID: PMC8512562
- DOI: 10.3390/molecules26196023
Semisynthetic Derivatives of Selected Amaryllidaceae Alkaloids as a New Class of Antimycobacterial Agents
Abstract
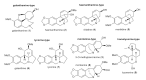
The search for novel antimycobacterial drugs is a matter of urgency, since tuberculosis is still one of the top ten causes of death from a single infectious agent, killing more than 1.4 million people worldwide each year. Nine Amaryllidaceae alkaloids (AAs) of various structural types have been screened for their antimycobacterial activity. Unfortunately, all were considered inactive, and thus a pilot series of aromatic esters of galanthamine, 3-O-methylpancracine, vittatine and maritidine were synthesized to increase biological activity. The semisynthetic derivatives of AAs were screened for their in vitro antimycobacterial activity against Mycobacterium tuberculosis H37Ra and two other mycobacterial strains (M. aurum, M. smegmatis) using a modified Microplate Alamar Blue Assay. The most active compounds were also studied for their in vitro hepatotoxicity on the hepatocellular carcinoma cell line HepG2. In general, the derivatization of the original AAs was associated with a significant increase in antimycobacterial activity. Several pilot derivatives were identified as compounds with micromolar MICs against M. tuberculosis H37Ra. Two derivatives of galanthamine, 1i and 1r, were selected for further structure optimalization to increase the selectivity index.
Keywords: 3-O-methylpancracine; Amaryllidaceae; analogues; antimycobacterial activity; cytotoxicity; galanthamine; tuberculosis.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
Alkaloids of Dicranostigma franchetianum (Papaveraceae) and Berberine Derivatives as a New Class of Antimycobacterial Agents.Biomolecules. 2022 Jun 17;12(6):844. doi: 10.3390/biom12060844. Biomolecules. 2022. PMID: 35740968 Free PMC article.
-
5-Alkylamino-N-phenylpyrazine-2-carboxamides: Design, Preparation, and Antimycobacterial Evaluation.Molecules. 2020 Mar 28;25(7):1561. doi: 10.3390/molecules25071561. Molecules. 2020. PMID: 32231166 Free PMC article.
-
N-Pyrazinoyl Substituted Amino Acids as Potential Antimycobacterial Agents-The Synthesis and Biological Evaluation of Enantiomers.Molecules. 2020 Mar 27;25(7):1518. doi: 10.3390/molecules25071518. Molecules. 2020. PMID: 32230728 Free PMC article.
-
[Development of antituberculous drugs: current status and future prospects].Kekkaku. 2006 Dec;81(12):753-74. Kekkaku. 2006. PMID: 17240921 Review. Japanese.
-
A mini-review of the anti-SARS-CoV-2 potency of Amaryllidaceae alkaloids.Phytomedicine. 2024 Jul;129:155576. doi: 10.1016/j.phymed.2024.155576. Epub 2024 Mar 29. Phytomedicine. 2024. PMID: 38579643 Review.
Cited by
-
Alkaloids of Dicranostigma franchetianum (Papaveraceae) and Berberine Derivatives as a New Class of Antimycobacterial Agents.Biomolecules. 2022 Jun 17;12(6):844. doi: 10.3390/biom12060844. Biomolecules. 2022. PMID: 35740968 Free PMC article.
-
Investigation of the antimycobacterial activity of African medicinal plants combined with chemometric analysis to identify potential leads.Sci Rep. 2024 Jun 25;14(1):14660. doi: 10.1038/s41598-024-65369-7. Sci Rep. 2024. PMID: 38918410 Free PMC article.
References
-
- Kanabalan R.D., Lee L.J., Lee T.Y., Chong P.P., Hassan L., Ismail R., Chin V.K. Human tuberculosis and Mycobacterium tuberculosis complex: A review on genetic diversity, pathogenesis and omics approaches in host biomarkers discovery. Microbiol. Res. 2021;246:126674. doi: 10.1016/j.micres.2020.126674. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

