Efficacy and Safety of Subcutaneous Infusion of Non-formulated Furosemide in Patients with Worsening Heart Failure: a Real-World Study
- PMID: 34642870
- PMCID: PMC9213343
- DOI: 10.1007/s12265-021-10173-1
Efficacy and Safety of Subcutaneous Infusion of Non-formulated Furosemide in Patients with Worsening Heart Failure: a Real-World Study
Abstract
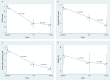
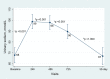
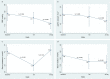
We aimed to evaluate the efficacy (short-term changes in surrogates of decongestion) and safety following the ambulatory administration of subcutaneous furosemide (SCF) in patients with WHF. Fifty-five ambulatory patients were treated with SCF administered by an elastomeric pump for at least 72 h. Surrogates of congestion were assessed at baseline, 72 h, and 30 days. Spot urinary sodium (uNa+) was assessed at baseline, 24-48-72 h, and 30 days. The median (IQI) of NT-proBNP and uNa+ at baseline was 5218 pg/mL (2856-10878) and 68±3 mmol/L, respectively. Following administration of SCF (median dose of 100 mg/daily), we found a sustained increase in uNa+ during the first 72 h of treatment compared to baseline, paralleled with evidence of decongestion at 72 h, and 30 days. No significant safety concerns were observed. SCF was an effective and safe diuretic strategy for outpatient congestion management. Non-formulated subcutaneous furosemide in patients with WHF. Efficacy and safety.
Keywords: Diuretics; Fluid overload; Subcutaneous furosemide; Urinary sodium; Worsening heart failure.
© 2021. The Author(s).
Conflict of interest statement
Rafael de la Espriella received board speaker fees and travel expenses from Novartis, Rovi, Pfizer, Daiichi Sankyo, Astra-Zeneca, NovoNordisk, Boehringer Ingelheim, Lilly. Gema Miñana received speaker fees and travel expenses from Abbott. Enrique Santas received board speaker fees and travel expenses from Novartis, Rovi, Pfizer, Daiichi Sankyo, Astra-Zeneca, NovoNordisk, Boehringer Ingelheim. Julio Núñez received board speaker fees and travel expenses from Novartis, Rovi, Pfizer, Daiichi Sankyo, Astra-Zeneca, NovoNordisk, Boehringer Ingelheim, Lilly. All other authors declare no competing interests.
Figures


References
-
- Ponikowski P, Voors AA, Anker SD, Bueno H, Cleland JGF, Coats AJS, Falk V, González-Juanatey JR, Harjola VP, Jankowska EA, Jessup M, Linde C, Nihoyannopoulos P, Parissis JT, Pieske B, Riley JP, Rosano GMC, Ruilope LM, Ruschitzka F, Rutten FH, van der Meer P, ESC Scientific Document Group 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: The Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC)Developed with the special contribution of the Heart Failure Association (HFA) of the ESC. European Heart Journal. 2016;37(27):2129–2200. doi: 10.1093/eurheartj/ehw128. - DOI - PubMed
-
- Mebazaa A, Yilmaz MB, Levy P, Ponikowski P, Peacock WF, Laribi S, Ristic AD, Lambrinou E, Masip J, Riley JP, McDonagh T, Mueller C, deFilippi C, Harjola VP, Thiele H, Piepoli MF, Metra M, Maggioni A, McMurray J, Dickstein K, Damman K, Seferovic PM, Ruschitzka F, Leite-Moreira AF, Bellou A, Anker SD. Filippatos G. Recommendations on pre-hospital & early hospital management of acute heart failure: A consensus paper from the Heart Failure Association of the European Society of Cardiology, the European Society of Emergency Medicine and the Society of Academic Emergency Medicine. European Journal of Heart Failure. 2015;17(6):544–558. doi: 10.1002/ejhf.289. - DOI - PubMed
-
- Wierda E, Dickhoff C, Handoko ML, Oosterom L, Kok WE, de Rover Y, de Mol BAJM, van Heerebeek L, Schroeder-Tanka JM. Outpatient treatment of worsening heart failure with intravenous and subcutaneous diuretics: A systematic review of the literature. ESC Heart Failure. 2020;7(3):892–902. doi: 10.1002/ehf2.12677. - DOI - PMC - PubMed
-
- Buckley LF, Carter DM, Matta L, Cheng JW, Stevens C, Belenkiy RM, Burpee LJ, Young MA, Weiffenbach CS, Smallwood JA, Stevenson LW, Desai AS. Intravenous diuretic therapy for the management of heart failure and volume overload in a multidisciplinary outpatient unit. JACC Heart Failure. 2016;4(1):1–8. doi: 10.1016/j.jchf.2015.06.017. - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

