Disordered breathing in a Pitt-Hopkins syndrome model involves Phox2b-expressing parafacial neurons and aberrant Nav1.8 expression
- PMID: 34645823
- PMCID: PMC8514575
- DOI: 10.1038/s41467-021-26263-2
Disordered breathing in a Pitt-Hopkins syndrome model involves Phox2b-expressing parafacial neurons and aberrant Nav1.8 expression
Abstract
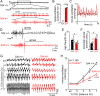
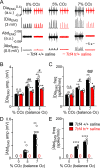
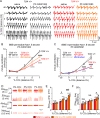
Pitt-Hopkins syndrome (PTHS) is a rare autism spectrum-like disorder characterized by intellectual disability, developmental delays, and breathing problems involving episodes of hyperventilation followed by apnea. PTHS is caused by functional haploinsufficiency of the gene encoding transcription factor 4 (Tcf4). Despite the severity of this disease, mechanisms contributing to PTHS behavioral abnormalities are not well understood. Here, we show that a Tcf4 truncation (Tcf4tr/+) mouse model of PTHS exhibits breathing problems similar to PTHS patients. This behavioral deficit is associated with selective loss of putative expiratory parafacial neurons and compromised function of neurons in the retrotrapezoid nucleus that regulate breathing in response to tissue CO2/H+. We also show that central Nav1.8 channels can be targeted pharmacologically to improve respiratory function at the cellular and behavioral levels in Tcf4tr/+ mice, thus establishing Nav1.8 as a high priority target with therapeutic potential in PTHS.
© 2021. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures






References
Publication types
MeSH terms
Substances
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

