The Treasury of Wharton's Jelly
- PMID: 34647276
- PMCID: PMC9209346
- DOI: 10.1007/s12015-021-10217-8
The Treasury of Wharton's Jelly
Abstract
Background: Postnatal umbilical cord tissue contains valuable mesenchymal progenitor cells of various differentiation stages. While mesenchymal stem cells are plastic-adherent and tend to differentiate into myofibroblastic phenotypes, some round cells detach, float above the adherent cells, and build up cell aggregates, or form spheroids spontaneously. Very small luminescent cells are always involved as single cells or within collective forms and resemble the common well-known very small embryonic-like cells (VSELs). In this study, we investigated these VSELs-like cells in terms of their pluripotency phenotype and tri-lineage differentiation potential.
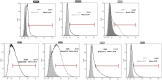
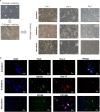
Methods: VSELs-like cells were isolated from cell-culture supernatants by a process that combines filtering, up concentration, and centrifugation. To determine their pluripotency character, we measured the expression of Nanog, Sox-2, Oct-4, SSEA-1, CXCR4, SSEA-4 on gene and protein level. In addition, the cultured cells derived from UC tissue were examined regarding their potential to differentiate into three germ layers.
Result: The VSELs-like cells express all of the pluripotency-associated markers we investigated and are able to differentiate into meso- endo- and ectodermal precursor cells.
Conclusions: Umbilical cord tissue hosts highly potent VSELs-like stem cells.
Keywords: Multipotency; Pluripotency; Regenerative medicine; Trilineage differentiation.; Umbilical cord tissue; Very small embryonic like stem cells; Wharton's jelly.
© 2021. The Author(s).
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
-
- Subramanian, A., Fong, C. Y., Biswas, A., & Bongso, A. (2015). Comparative characterization of cells from the various compartments of the human umbilical cord shows that the Wharton’s jelly compartment provides the best source of clinically utilizable mesenchymal stem cells. PLoS One, 10(6). 10.1371/journal.pone.0127992.e0127992 - PMC - PubMed
-
- Enzmann, H., & Daniel, V. (1991). Die Diagnose des “excited-skin-syndrome” aus dem Blut. Eine Hilfe für die plastische Chirurgie. Laryngo- Rhino- Otologie,70(4), S. 184-186. 10.1055/s-2007-998017 - PubMed
-
- Stolzing, A., Jones, E., McGonagle, D., & Scutt, A. (2008). Age-related changes in human bone marrow-derived mesenchymal stem cells: consequences for cell therapies. Mechanisms of Ageing and Development,129(3), S. 163-173. 10.1016/j.mad.2007.12.002 - PubMed
-
- Khan, H., Mafi, P., Mafi, R., & Khan, W. (2018). The effects of ageing on differentiation and characterisation of human mesenchymal stem cells. Current Stem Cell Research & Therapy,13(5), S. 378-383. 10.2174/1574888X11666160429122527 - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

