Design and reporting characteristics of clinical trials investigating sedation practices in the paediatric intensive care unit: a scoping review by SCEPTER (Sedation Consortium on Endpoints and Procedures for Treatment, Education and Research)
- PMID: 34649849
- PMCID: PMC8522672
- DOI: 10.1136/bmjopen-2021-053519
Design and reporting characteristics of clinical trials investigating sedation practices in the paediatric intensive care unit: a scoping review by SCEPTER (Sedation Consortium on Endpoints and Procedures for Treatment, Education and Research)
Abstract
Objectives: To conduct a scoping review of sedation clinical trials in the paediatric intensive care setting and summarise key methodological elements.
Design: Scoping review.
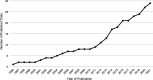
Data sources: PubMed, Embase, Cumulative Index to Nursing and Allied Health Literature and grey references including ClinicalTrials.gov from database inception to 3 August 2021.
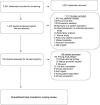
Study selection: All human trials in the English language related to sedation in paediatric critically ill patients were included. After title and abstract screening, full-text review was performed. 29 trials were eligible for final analysis.
Data extraction: A coding manual was developed and pretested. Trial characteristics were double extracted.
Results: The majority of trials were single centre (22/29, 75.9%), parallel group superiority (17/29, 58.6%), double-blinded (18/29, 62.1%) and conducted in an academic setting (29/29, 100.0%). Trial enrolment (≥90% planned sample size) was achieved in 65.5% of trials (19/29), and retention (≥90% enrolled subjects) in 72.4% of trials (21/29). Protocol violations were reported in nine trials (31.0%). The most commonly studied cohorts were mechanically ventilated patients (28/29, 96.6%) and postsurgical patients (11/29, 37.9%) with inclusion criteria for age ranging from 0±0.5 to 15.0±7.3 years (median±IQR). The median age of enrolled patients was 1.7 years (IQR=4.4 years). Patients excluded from trials were those with neurological impairment (21/29, 72.4%), complex disease (20/29, 69.0%) or receipt of neuromuscular blockade (10/29, 34.5%). Trials evaluated drugs/protocols for sedation management (20/29, 69.0%), weaning (3/29, 10.3%), daily interruption (3/29, 10.3%) or protocolisation (3/29, 10.3%). Primary outcome measures were heterogeneous, as were assessment instruments and follow-up durations.
Conclusions: There is substantial heterogeneity in methodological approach in clinical trials evaluating sedation in critically ill paediatric patients. These results provide a basis for the design of future clinical trials to improve the quality of trial data and aid in the development of sedation-related clinical guidelines.
Keywords: clinical trials; paediatric intensive & critical care; protocols & guidelines; statistics & research methods.
© Author(s) (or their employer(s)) 2021. Re-use permitted under CC BY-NC. No commercial re-use. See rights and permissions. Published by BMJ.
Conflict of interest statement
Competing interests: None declared.
Figures
Similar articles
-
Protocol-directed sedation versus non-protocol-directed sedation in mechanically ventilated intensive care adults and children.Cochrane Database Syst Rev. 2018 Nov 12;11(11):CD009771. doi: 10.1002/14651858.CD009771.pub3. Cochrane Database Syst Rev. 2018. PMID: 30480753 Free PMC article.
-
Routine gastric residual volume measurement to guide enteral feeding in mechanically ventilated infants and children: the GASTRIC feasibility study.Health Technol Assess. 2020 May;24(23):1-120. doi: 10.3310/hta24230. Health Technol Assess. 2020. PMID: 32458797 Free PMC article. Clinical Trial.
-
Protocolized sedation vs usual care in pediatric patients mechanically ventilated for acute respiratory failure: a randomized clinical trial.JAMA. 2015 Jan 27;313(4):379-89. doi: 10.1001/jama.2014.18399. JAMA. 2015. PMID: 25602358 Free PMC article. Clinical Trial.
-
Implementing early rehabilitation and mobilisation for children in UK paediatric intensive care units: the PERMIT feasibility study.Health Technol Assess. 2023 Nov;27(27):1-155. doi: 10.3310/HYRW5688. Health Technol Assess. 2023. PMID: 38063184 Free PMC article.
-
Psychometric analysis of subjective sedation scales used for critically ill paediatric patients.Nurs Crit Care. 2018 Jan;23(1):30-41. doi: 10.1111/nicc.12325. Epub 2017 Nov 12. Nurs Crit Care. 2018. PMID: 29131465 Review.
References
-
- Harris J, Ramelet A-S, van Dijk M, et al. . Clinical recommendations for pain, sedation, withdrawal and delirium assessment in critically ill infants and children: an ESPNIC position statement for healthcare professionals. Intensive Care Med 2016;42:972–86. 10.1007/s00134-016-4344-1 - DOI - PMC - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
