Ketamine increases activity of a fronto-striatal projection that regulates compulsive behavior in SAPAP3 knockout mice
- PMID: 34654803
- PMCID: PMC8519915
- DOI: 10.1038/s41467-021-26247-2
Ketamine increases activity of a fronto-striatal projection that regulates compulsive behavior in SAPAP3 knockout mice
Abstract
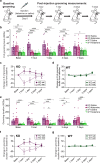
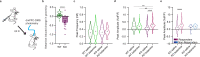
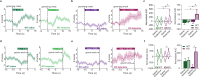
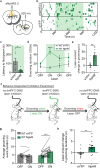
Obsessive-Compulsive Disorder (OCD), characterized by intrusive thoughts (obsessions) and repetitive behaviors (compulsions), is associated with dysfunction in fronto-striatal circuits. There are currently no fast-acting pharmacological treatments for OCD. However, recent clinical studies demonstrated that an intravenous infusion of ketamine rapidly reduces OCD symptoms. To probe mechanisms underlying ketamine's therapeutic effect on OCD-like behaviors, we used the SAPAP3 knockout (KO) mouse model of compulsive grooming. Here we recapitulate the fast-acting therapeutic effect of ketamine on compulsive behavior, and show that ketamine increases activity of dorsomedial prefrontal neurons projecting to the dorsomedial striatum in KO mice. Optogenetically mimicking this increase in fronto-striatal activity reduced compulsive grooming behavior in KO mice. Conversely, inhibiting this circuit in wild-type mice increased grooming. Finally, we demonstrate that ketamine blocks the exacerbation of grooming in KO mice caused by optogenetically inhibiting fronto-striatal activity. These studies demonstrate that ketamine increases activity in a fronto-striatal circuit that causally controls compulsive grooming behavior, suggesting this circuit may be important for ketamine's therapeutic effects in OCD.
© 2021. The Author(s).
Conflict of interest statement
In the last 3 years, Dr. Rodriguez has served as a consultant for Epiodyne and received research grant support from Biohaven Pharmaceuticals and a stipend from APA Publishing for her role as Deputy Editor at The American Journal of Psychiatry. All other authors report no financial or other relationships relevant to the subject of this manuscript.
Figures



Similar articles
-
Optogenetic inhibition of indirect pathway neurons in the dorsomedial striatum reduces excessive grooming in Sapap3-knockout mice.Neuropsychopharmacology. 2022 Jan;47(2):477-487. doi: 10.1038/s41386-021-01161-9. Epub 2021 Aug 20. Neuropsychopharmacology. 2022. PMID: 34417544 Free PMC article.
-
Early-life social isolation stress worsens compulsive- and anxiety-like behaviour and alters gene expression in the prefrontal cortex of SAPAP3 knockout mice.J Affect Disord. 2025 Nov 1;388:119600. doi: 10.1016/j.jad.2025.119600. Epub 2025 Jun 4. J Affect Disord. 2025. PMID: 40480392
-
Increased Metabotropic Glutamate Receptor 5 Signaling Underlies Obsessive-Compulsive Disorder-like Behavioral and Striatal Circuit Abnormalities in Mice.Biol Psychiatry. 2016 Oct 1;80(7):522-33. doi: 10.1016/j.biopsych.2016.04.023. Epub 2016 May 13. Biol Psychiatry. 2016. PMID: 27436084 Free PMC article.
-
Molecular and cellular basis of obsessive-compulsive disorder-like behaviors: emerging view from mouse models.Curr Opin Neurol. 2011 Apr;24(2):114-8. doi: 10.1097/WCO.0b013e32834451fb. Curr Opin Neurol. 2011. PMID: 21386675 Review.
-
Obsessive-compulsive disorder: beyond segregated cortico-striatal pathways.Trends Cogn Sci. 2012 Jan;16(1):43-51. doi: 10.1016/j.tics.2011.11.003. Epub 2011 Dec 2. Trends Cogn Sci. 2012. PMID: 22138231 Free PMC article. Review.
Cited by
-
Striking Long Term Beneficial Effects of Single Dose Psilocybin and Psychedelic Mushroom Extract in the SAPAP3 Rodent Model of OCD-Like Excessive Self-Grooming.bioRxiv [Preprint]. 2024 Jun 29:2024.06.25.600634. doi: 10.1101/2024.06.25.600634. bioRxiv. 2024. Update in: Mol Psychiatry. 2025 Mar;30(3):1172-1183. doi: 10.1038/s41380-024-02786-0. PMID: 38979304 Free PMC article. Updated. Preprint.
-
SAPAP Scaffold Proteins: From Synaptic Function to Neuropsychiatric Disorders.Cells. 2022 Nov 28;11(23):3815. doi: 10.3390/cells11233815. Cells. 2022. PMID: 36497075 Free PMC article. Review.
-
Unraveling the mechanisms of deep-brain stimulation of the internal capsule in a mouse model.Nat Commun. 2023 Sep 4;14(1):5385. doi: 10.1038/s41467-023-41026-x. Nat Commun. 2023. PMID: 37666830 Free PMC article.
-
Closed-loop recruitment of striatal interneurons prevents compulsive-like grooming behaviors.Nat Neurosci. 2024 Jun;27(6):1148-1156. doi: 10.1038/s41593-024-01633-3. Epub 2024 May 1. Nat Neurosci. 2024. PMID: 38693349 Free PMC article.
-
Striking long-term beneficial effects of single dose psilocybin and psychedelic mushroom extract in the SAPAP3 rodent model of OCD-like excessive self-grooming.Mol Psychiatry. 2025 Mar;30(3):1172-1183. doi: 10.1038/s41380-024-02786-0. Epub 2024 Oct 11. Mol Psychiatry. 2025. PMID: 39394457 Free PMC article.

