Neuro-immune-metabolism: The tripod system of homeostasis
- PMID: 34655659
- PMCID: PMC7611970
- DOI: 10.1016/j.imlet.2021.10.001
Neuro-immune-metabolism: The tripod system of homeostasis
Abstract
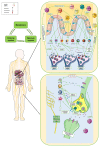
Homeostatic regulation of cellular and molecular processes is essential for the efficient physiological functioning of body organs. It requires an intricate balance of several networks throughout the body, most notable being the nervous, immune and metabolic systems. Several studies have reported the interactions between neuro-immune, immune-metabolic and neuro-metabolic pathways. Current review aims to integrate the information and show that neuro, immune and metabolic systems form the triumvirate of homeostasis. It focuses on the cellular and molecular interactions occurring in the extremities and intestine, which are innervated by the peripheral nervous system and for the intestine in particular the enteric nervous system. While the interdependence of neuro-immune-metabolic pathways provides a fallback mechanism in case of disruption of homeostasis, in chronic pathologies of continued disequilibrium, the collapse of one system spreads to the other interacting networks as well. Current review illustrates this domino-effect using diabetes as the main example. Together, this review attempts to provide a holistic picture of the integrated network of neuro-immune-metabolism and attempts to broaden the outlook when devising a scientific study or a treatment strategy.
Keywords: Diabetes; Homeostasis; Immune-metabolic interactions; Neuro-immune interactions; Neuro-metabolic interactions; Obesity; Peripheral nervous system.
Copyright © 2021. Published by Elsevier B.V.
Conflict of interest statement
Although unrelated to this article, L.F. is an employee of F. Hoffmann - La Roche, Basel now.
Figures

References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

