Fenton/Fenton-like metal-based nanomaterials combine with oxidase for synergistic tumor therapy
- PMID: 34656118
- PMCID: PMC8520258
- DOI: 10.1186/s12951-021-01074-1
Fenton/Fenton-like metal-based nanomaterials combine with oxidase for synergistic tumor therapy
Abstract
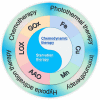
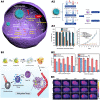
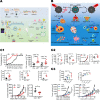
Chemodynamic therapy (CDT) catalyzed by transition metal and starvation therapy catalyzed by intracellular metabolite oxidases are both classic tumor treatments based on nanocatalysts. CDT monotherapy has limitations including low catalytic efficiency of metal ions and insufficient endogenous hydrogen peroxide (H2O2). Also, single starvation therapy shows limited ability on resisting tumors. The "metal-oxidase" cascade catalytic system is to introduce intracellular metabolite oxidases into the metal-based nanoplatform, which perfectly solves the shortcomings of the above-mentioned monotherapiesIn this system, oxidases can not only consume tumor nutrients to produce a "starvation effect", but also provide CDT with sufficient H2O2 and a suitable acidic environment, which further promote synergy between CDT and starvation therapy, leading to enhanced antitumor effects. More importantly, the "metal-oxidase" system can be combined with other antitumor therapies (such as photothermal therapy, hypoxia-activated drug therapy, chemotherapy, and immunotherapy) to maximize their antitumor effects. In addition, both metal-based nanoparticles and oxidases can activate tumor immunity through multiple pathways, so the combination of the "metal-oxidase" system with immunotherapy has a powerful synergistic effect. This article firstly introduced the metals which induce CDT and the oxidases which induce starvation therapy and then described the "metal-oxidase" cascade catalytic system in detail. Moreover, we highlight the application of the "metal-oxidase" system in combination with numerous antitumor therapies, especially in combination with immunotherapy, expecting to provide new ideas for tumor treatment.
Keywords: Chemodynamic therapy; Fenton reaction; Immunotherapy; Metabolite oxidase; Tumor synergistic therapy.
© 2021. The Author(s).
Conflict of interest statement
The authors declare that they have no competing interests.
Figures









Similar articles
-
A multivalent polyphenol-metal-nanoplatform for cascade amplified chemo-chemodynamic therapy.Acta Biomater. 2024 Jan 1;173:389-402. doi: 10.1016/j.actbio.2023.11.006. Epub 2023 Nov 14. Acta Biomater. 2024. PMID: 37967695
-
Pillar[6]arene-Based Supramolecular Nanocatalysts for Synergistically Enhanced Chemodynamic Therapy by the Intracellular Cascade Reaction.ACS Appl Mater Interfaces. 2021 Nov 17;13(45):53574-53585. doi: 10.1021/acsami.1c15203. Epub 2021 Nov 3. ACS Appl Mater Interfaces. 2021. PMID: 34729975
-
Phosphate-Degradable Nanoparticles Based on Metal-Organic Frameworks for Chemo-Starvation-Chemodynamic Synergistic Antitumor Therapy.ACS Appl Mater Interfaces. 2021 Aug 11;13(31):37713-37723. doi: 10.1021/acsami.1c10816. Epub 2021 Aug 3. ACS Appl Mater Interfaces. 2021. PMID: 34340302
-
Recent advances in multifunctional nanomaterials for photothermal-enhanced Fenton-based chemodynamic tumor therapy.Mater Today Bio. 2022 Jan 4;13:100197. doi: 10.1016/j.mtbio.2021.100197. eCollection 2022 Jan. Mater Today Bio. 2022. PMID: 35036895 Free PMC article. Review.
-
Chemodynamic Therapy via Fenton and Fenton-Like Nanomaterials: Strategies and Recent Advances.Small. 2022 Feb;18(6):e2103868. doi: 10.1002/smll.202103868. Epub 2021 Nov 2. Small. 2022. PMID: 34729913 Review.
Cited by
-
Current Advances on Nanomaterials Interfering with Lactate Metabolism for Tumor Therapy.Adv Sci (Weinh). 2024 Jan;11(3):e2305662. doi: 10.1002/advs.202305662. Epub 2023 Nov 8. Adv Sci (Weinh). 2024. PMID: 37941489 Free PMC article. Review.
-
Antioxidant-related enzymes and peptides as biomarkers of metallic nanoparticles (eco)toxicity in the aquatic environment.Chemosphere. 2024 Sep;364:142988. doi: 10.1016/j.chemosphere.2024.142988. Epub 2024 Aug 3. Chemosphere. 2024. PMID: 39103097 Free PMC article. Review.
-
Multi-Stimuli-Responsive Tadpole-like Polymer/Lipid Janus Microrobots for Advanced Smart Material Applications.ACS Appl Mater Interfaces. 2024 Mar 27;16(12):15533-15547. doi: 10.1021/acsami.3c18826. Epub 2024 Feb 15. ACS Appl Mater Interfaces. 2024. PMID: 38356451 Free PMC article.
-
Engineering the glioblastoma microenvironment with bioactive nanoparticles for effective immunotherapy.RSC Adv. 2023 Oct 27;13(45):31411-31425. doi: 10.1039/d3ra01153d. eCollection 2023 Oct 26. RSC Adv. 2023. PMID: 37901257 Free PMC article. Review.
-
Novel adjuvant delivery system constructed by alum-emulsion hybrid nanoparticles with TLR9 agonists boosts vaccine immunity.J Nanobiotechnology. 2025 Jul 1;23(1):472. doi: 10.1186/s12951-025-03560-2. J Nanobiotechnology. 2025. PMID: 40598254 Free PMC article.
References
-
- Fidler MM, Gupta S, Soerjomataram I, Ferlay J, Steliarova-Foucher E, Bray F. Cancer incidence and mortality among young adults aged 20–39 years worldwide in 2012: a population-based study. Lancet Oncol. 2017;18:1579–1589. - PubMed
-
- Wei W, Zeng H, Zheng R, Zhang S, An L, Chen R, et al. Cancer registration in China and its role in cancer prevention and control. Lancet Oncol. 2020;21:e342–e349. - PubMed
-
- Chen W, Zheng R, Baade PD, Zhang S, Zeng H, Bray F, et al. Cancer statistics in China, 2015. CA Cancer J Clin. 2016;66:115–132. - PubMed
-
- Alexis F, Rhee JW, Richie JP, Radovic-Moreno AF, Langer R, Farokhzad OC. New frontiers in nanotechnology for cancer treatment. Urol Oncol. 2008;26:74–85. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

