Review of Scavenged Sampling for Sustainable Therapeutic Drug Monitoring: Do More With Less
- PMID: 34657093
- PMCID: PMC8746914
- DOI: 10.1097/FTD.0000000000000928
Review of Scavenged Sampling for Sustainable Therapeutic Drug Monitoring: Do More With Less
Abstract
Purpose: Innovative and sustainable sampling strategies for bioanalytical quantification of drugs and metabolites have gained considerable interest. Scavenging can be stratified as a sustainable sampling strategy using residual material because it aligns with the green principles of waste reduction and sampling optimization. Scavenged sampling includes all biological fluids' (eg, blood, liquor, and urine) leftover from standard clinical care. This review elaborates on the past and current landscape of sustainable sampling within therapeutic drug monitoring, with a focus on scavenged sampling.
Methods: In February 2021, 4 databases were searched to assess the literature on the clinical use of innovative and sustainable sampling techniques without applying publication date restrictions. Studies reporting the clinical use of scavenged blood sampling and bridging studies of scavenged sampling and normal blood sampling were eligible for inclusion.
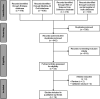
Results: Overall, 19 eligible studies concerning scavenged sampling were identified from 1441 records. Scavenged sampling is mainly applied in the pediatric population, although other patient groups may benefit from this strategy. The infrastructure required for scavenged sampling encounters several challenges, including logistic hurdles, storage and handling conditions, and documentation errors. A workflow is proposed with identified opportunities that guide the implementation of scavenged sampling.
Conclusions: This review presents current evidence on the clinical use of scavenged sampling strategies. Scavenged sampling can be a suitable approach for drug quantification to improve dosage regimens, perform pharmacokinetic studies, and explore the value of therapeutic drug monitoring without additional sample collection.
Copyright © 2021 The Author(s). Published by Wolters Kluwer Health, Inc. on behalf of the International Association of Therapeutic Drug Monitoring and Clinical Toxicology.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
-
- Le J, Bradley JS. Optimizing antibiotic drug therapy in pediatrics: current state and future needs. J Clin Pharmacol. 2018;58(suppl 10):S108–S122. - PubMed
-
- Hensher M, McGain F. Health care sustainability metrics: building a safer, low-carbon health system. Health Aff (Millwood). 2020;39:2080–2087. - PubMed
-
- Kleber J, Cohen B. Reducing waste and increasing sustainability in health care settings. Am J Nurs. 2020;120:45–48. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Miscellaneous

