Mapping input noise to escape noise in integrate-and-fire neurons: a level-crossing approach
- PMID: 34668051
- PMCID: PMC8551127
- DOI: 10.1007/s00422-021-00899-1
Mapping input noise to escape noise in integrate-and-fire neurons: a level-crossing approach
Abstract
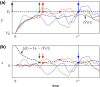
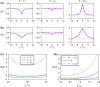
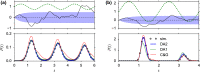
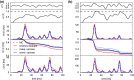
Noise in spiking neurons is commonly modeled by a noisy input current or by generating output spikes stochastically with a voltage-dependent hazard rate ("escape noise"). While input noise lends itself to modeling biophysical noise processes, the phenomenological escape noise is mathematically more tractable. Using the level-crossing theory for differentiable Gaussian processes, we derive an approximate mapping between colored input noise and escape noise in leaky integrate-and-fire neurons. This mapping requires the first-passage-time (FPT) density of an overdamped Brownian particle driven by colored noise with respect to an arbitrarily moving boundary. Starting from the Wiener-Rice series for the FPT density, we apply the second-order decoupling approximation of Stratonovich to the case of moving boundaries and derive a simplified hazard-rate representation that is local in time and numerically efficient. This simplification requires the calculation of the non-stationary auto-correlation function of the level-crossing process: For exponentially correlated input noise (Ornstein-Uhlenbeck process), we obtain an exact formula for the zero-lag auto-correlation as a function of noise parameters, mean membrane potential and its speed, as well as an exponential approximation of the full auto-correlation function. The theory well predicts the FPT and interspike interval densities as well as the population activities obtained from simulations with colored input noise and time-dependent stimulus or boundary. The agreement with simulations is strongly enhanced across the sub- and suprathreshold firing regime compared to a first-order decoupling approximation that neglects correlations between level crossings. The second-order approximation also improves upon a previously proposed theory in the subthreshold regime. Depending on a simplicity-accuracy trade-off, all considered approximations represent useful mappings from colored input noise to escape noise, enabling progress in the theory of neuronal population dynamics.
Keywords: Colored noise; Escape noise; First-passage-time density; Hazard rate; Integrate-and-fire neuron; Interspike interval density; Neuronal population dynamics; Threshold-crossing statistics.
© 2021. The Author(s).
Conflict of interest statement
The author declares that he has no conflict of interest.
Figures
Similar articles
-
Exact analytical results for integrate-and-fire neurons driven by excitatory shot noise.J Comput Neurosci. 2017 Aug;43(1):81-91. doi: 10.1007/s10827-017-0649-5. Epub 2017 Jun 6. J Comput Neurosci. 2017. PMID: 28585050
-
Integrate-and-fire neurons driven by asymmetric dichotomous noise.Biol Cybern. 2014 Dec;108(6):825-43. doi: 10.1007/s00422-014-0621-7. Epub 2014 Jul 19. Biol Cybern. 2014. PMID: 25037240
-
Noise in integrate-and-fire neurons: from stochastic input to escape rates.Neural Comput. 2000 Feb;12(2):367-84. doi: 10.1162/089976600300015835. Neural Comput. 2000. PMID: 10636947
-
Mean-return-time phase of a stochastic oscillator provides an approximate renewal description for the associated point process.Biol Cybern. 2022 Apr;116(2):235-251. doi: 10.1007/s00422-022-00920-1. Epub 2022 Feb 15. Biol Cybern. 2022. PMID: 35166932 Free PMC article. Review.
-
The stochastic diffusion models of nerve membrane depolarization and interspike interval generation.J Peripher Nerv Syst. 1999;4(1):27-42. J Peripher Nerv Syst. 1999. PMID: 10197063 Review.
Cited by
-
A refractory density approach to a multi-scale SEIRS epidemic model.Infect Dis Model. 2025 Mar 13;10(3):787-801. doi: 10.1016/j.idm.2025.03.004. eCollection 2025 Sep. Infect Dis Model. 2025. PMID: 40213022 Free PMC article.
-
Intra-ripple frequency accommodation in an inhibitory network model for hippocampal ripple oscillations.PLoS Comput Biol. 2024 Feb 20;20(2):e1011886. doi: 10.1371/journal.pcbi.1011886. eCollection 2024 Feb. PLoS Comput Biol. 2024. PMID: 38377147 Free PMC article.
-
Solving Max-Cut Problem Using Spiking Boltzmann Machine Based on Neuromorphic Hardware with Phase Change Memory.Adv Sci (Weinh). 2024 Dec;11(46):e2406433. doi: 10.1002/advs.202406433. Epub 2024 Oct 23. Adv Sci (Weinh). 2024. PMID: 39440674 Free PMC article.
References
-
- Azaïs JM, Wschebor M. Level sets and extrema of random processes and fields. New Jersey: Wiley; 2009.
MeSH terms
LinkOut - more resources
Full Text Sources

