Cancer-Associated Fibroblast Subpopulations With Diverse and Dynamic Roles in the Tumor Microenvironment
- PMID: 34670861
- PMCID: PMC9306405
- DOI: 10.1158/1541-7786.MCR-21-0282
Cancer-Associated Fibroblast Subpopulations With Diverse and Dynamic Roles in the Tumor Microenvironment
Abstract
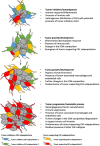
Close interactions between cancer cells and cancer-associated fibroblasts (CAF) have repeatedly been reported to support tumor progression. Yet, targeting CAFs has so far failed to show a real benefit in cancer treatment, as preclinical studies have shown that such a strategy can enhance tumor growth. Accordingly, recent paradigm-shifting data suggest that certain CAF subpopulations could also show tumor-inhibitory capabilities. The present review aims to provide an in-depth description of the cellular heterogeneity of the CAF compartment in tumors. Through combining information from different cancer types, here we define 4 main CAF subpopulations that might cohabitate in any tumor microenvironment (TME). In addition, a model for the evolution of CAFs during tumor development is introduced. Moreover, the presence of tumor-inhibitory CAFs in the TME as well as their molecular characteristics are extensively discussed. Finally, the potential cellular origins of these distinct CAF subpopulations are reviewed. To our knowledge, this is the first attempt at establishing a broad but comprehensive classification of CAF subpopulations. Altogether, the present manuscript aims to provide the latest developments and innovative insights that could help refine future therapeutic targeting of CAFs for cancer treatment.
©2021 The Authors; Published by the American Association for Cancer Research.
Figures



References
-
- Simon T, Coquerel B, Petit A, Kassim Y, Demange E, Cerf D, et al. Direct effect of bevacizumab on glioblastoma cell lines in vitro. NeuroMol Med 2014;16:752–71. - PubMed
-
- Simon T, Gagliano T, Giamas G. Direct effects of anti-angiogenic therapies on tumor cells: VEGF signaling. Trends Mol Med 2017;23:282–92. - PubMed
-
- Liao Z, Tan ZW, Zhu P, Tan NS. Cancer-associated fibroblasts in tumor microenvironment - accomplices in tumor malignancy. Cell Immunol 2019;343:103729. - PubMed
-
- Brandao M, Simon T, Critchley G, Giamas G. Astrocytes, the rising stars of the glioblastoma microenvironment. Glia 2019;67:779–90. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources

