Induction of High Levels of Specific Humoral and Cellular Responses to SARS-CoV-2 After the Administration of Covid-19 mRNA Vaccines Requires Several Days
- PMID: 34671348
- PMCID: PMC8521189
- DOI: 10.3389/fimmu.2021.726960
Induction of High Levels of Specific Humoral and Cellular Responses to SARS-CoV-2 After the Administration of Covid-19 mRNA Vaccines Requires Several Days
Abstract
Objectives: In the context of the Covid-19 pandemic, the fast development of vaccines with efficacy of around 95% preventing Covid-19 illness provides a unique opportunity to reduce the mortality associated with the pandemic. However, in the absence of efficacious prophylactic medications and few treatments for this infection, the induction of a fast and robust protective immunity is required for effective disease control, not only to prevent the disease but also the infection and shedding/transmission. The objective of our study was to analyze the level of specific humoral and cellular T-cell responses against the spike protein of SARS-CoV-2 induced by two mRNA-based vaccines (BNT162b2 and mRNA-1273), but also how long it takes after vaccination to induce these protective humoral and cellular immune responses.
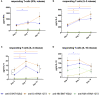
Methods: We studied in 40 healthy (not previously infected) volunteers vaccinated with BNT162b2 or mRNA-1273 vaccines the presence of spike-specific IgG antibodies and SARS-CoV-2-specific T cells at 3, 7 and 14 days after receiving the second dose of the vaccine. The specific T-cell response was analyzed stimulating fresh whole blood from vaccinated volunteers with SARS-CoV-2 peptides and measuring the release of cytokines secreted by T cells in response to SARS-CoV-2 stimulation.
Results: Our results indicate that the immunization capacity of both vaccines is comparable. However, although both BNT162b2 and mRNA-1273 vaccines can induce early B-cell and T-cell responses, these vaccine-mediated immune responses do not reach their maximum values until 14 days after completing the vaccination schedule.
Conclusion: This refractory period in the induction of specific immunity observed after completing the vaccination could constitute a window of higher infection risk, which could explain some emerging cases of SARS-CoV-2 infection in vaccinated people.
Keywords: COVID-19; SARS – CoV – 2; mRNA-vaccines; specific T-cell response; specific humoral response.
Copyright © 2021 Gil-Manso, Carbonell, López-Fernández, Miguens, Alonso, Buño, Muñoz, Ochando, Pion and Correa-Rocha.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

References
-
- WHO . Draft Landscape of COVID-19 Candidate Vaccines. Available at: https://www.who.int/publications/m/item/draft-landscape-of-covid-19-cand....
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

