Brain cerebrospinal fluid flow
- PMID: 34671708
- PMCID: PMC8525801
- DOI: 10.1103/physrevfluids.6.070501
Brain cerebrospinal fluid flow
Abstract
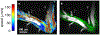
Cerebrospinal fluid flows around and into the brain, driven by intricate mechanisms, with profound implications for human health. According to the glymphatic hypothesis, in physiological conditions, cerebrospinal fluid flows primarily during sleep and serves to remove metabolic wastes like the amyloid-beta and tau proteins whose accumulation is believed to cause Alzheimer's disease. This paper reviews one research team's recent in vivo experiments and theoretical studies to better understand the fluid dynamics of brain cerebrospinal fluid flow. Driving mechanisms are considered, particularly arterial pulsation. Flow correlates closely with artery motion and changes when artery motion is manipulated. Though there are discrepancies between in vivo observations and predictions from simulations and theoretical studies of the mechanism, realistic boundary conditions bring closer agreement. Vessel shapes are considered, and have elongation that minimizes their hydraulic resistance, perhaps through evolutionary optimization. The pathological condition of stroke is considered. Much tissue damage after stroke is caused by swelling, and there is now strong evidence that early swelling is caused not by fluid from blood, as is commonly thought, but by cerebrospinal fluid. Finally, drug delivery is considered, and demonstrations show the glymphatic system could quickly deliver drugs across the blood-brain barrier. The paper closes with a discussion of future opportunities in the fast-changing field of brain fluid dynamics.
Figures

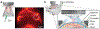
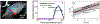



Similar articles
-
Perivascular pumping in the mouse brain: Improved boundary conditions reconcile theory, simulation, and experiment.J Theor Biol. 2022 Jun 7;542:111103. doi: 10.1016/j.jtbi.2022.111103. Epub 2022 Mar 23. J Theor Biol. 2022. PMID: 35339513 Free PMC article.
-
Cerebrospinal Fluid Flow.Annu Rev Fluid Mech. 2023;55:237-264. doi: 10.1146/annurev-fluid-120720-011638. Epub 2022 Sep 28. Annu Rev Fluid Mech. 2023. PMID: 39691763 Free PMC article.
-
Detection of Glymphatic Outflow of Tau from Brain to Cerebrospinal Fluid in Mice.Methods Mol Biol. 2024;2754:351-359. doi: 10.1007/978-1-0716-3629-9_19. Methods Mol Biol. 2024. PMID: 38512676
-
The impact of neurovascular, blood-brain barrier, and glymphatic dysfunction in neurodegenerative and metabolic diseases.Int Rev Neurobiol. 2020;154:413-436. doi: 10.1016/bs.irn.2020.02.006. Epub 2020 Jul 14. Int Rev Neurobiol. 2020. PMID: 32739013 Review.
-
The glymphatic pathway in neurological disorders.Lancet Neurol. 2018 Nov;17(11):1016-1024. doi: 10.1016/S1474-4422(18)30318-1. Lancet Neurol. 2018. PMID: 30353860 Free PMC article. Review.
Cited by
-
Fluid dynamics model of the cerebral ventricular system.Proc Natl Acad Sci U S A. 2025 Jul;122(26):e2426067122. doi: 10.1073/pnas.2426067122. Epub 2025 Jun 25. Proc Natl Acad Sci U S A. 2025. PMID: 40560615
-
Sleep, Glial Function, and the Endocannabinoid System: Implications for Neuroinflammation and Sleep Disorders.Int J Mol Sci. 2024 Mar 9;25(6):3160. doi: 10.3390/ijms25063160. Int J Mol Sci. 2024. PMID: 38542134 Free PMC article. Review.
-
The aging human brain exhibits reduced cerebrospinal fluid flow during sleep due to both neural and vascular factors.bioRxiv [Preprint]. 2025 Feb 24:2025.02.22.639649. doi: 10.1101/2025.02.22.639649. bioRxiv. 2025. PMID: 40060413 Free PMC article. Preprint.
-
Microenvironments Matter: Advances in Brain-on-Chip.Biosensors (Basel). 2023 May 16;13(5):551. doi: 10.3390/bios13050551. Biosensors (Basel). 2023. PMID: 37232912 Free PMC article. Review.
-
Age- and time-of-day dependence of glymphatic function in the human brain measured via two diffusion MRI methods.Front Aging Neurosci. 2023 May 22;15:1173221. doi: 10.3389/fnagi.2023.1173221. eCollection 2023. Front Aging Neurosci. 2023. PMID: 37284019 Free PMC article.
References
-
- Gaigalas Adolfas K, Hubbard Joseph B, McCurley M, and Woo Sam, “Diffusion of bovine serum albumin in aqueous solutions,” The Journal of Physical Chemistry 96, 2355–2359 (1992).
-
- Eide Per Kristian, Vinje Vegard, Pripp Are Hugo, Mardal Kent-André, and Ringstad Geir, “Sleep deprivation impairs molecular clearance from the human brain,” Brain 144, 863–874 (2021). - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
