Impact of neurodegenerative diseases on human adult hippocampal neurogenesis
- PMID: 34672693
- PMCID: PMC7613437
- DOI: 10.1126/science.abl5163
Impact of neurodegenerative diseases on human adult hippocampal neurogenesis
Abstract
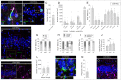
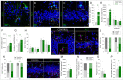
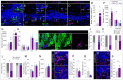
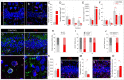
Disrupted hippocampal performance underlies psychiatric comorbidities and cognitive impairments in patients with neurodegenerative disorders. To understand the contribution of adult hippocampal neurogenesis (AHN) to amyotrophic lateral sclerosis, Huntington’s disease, Parkinson’s disease, dementia with Lewy bodies, and frontotemporal dementia, we studied postmortem human samples. We found that adult-born dentate granule cells showed abnormal morphological development and changes in the expression of differentiation markers. The ratio of quiescent to proliferating hippocampal neural stem cells shifted, and the homeostasis of the neurogenic niche was altered. Aging and neurodegenerative diseases reduced the phagocytic capacity of microglia, triggered astrogliosis, and altered the microvasculature of the dentate gyrus. Thus, enhanced vulnerability of AHN to neurodegeneration might underlie hippocampal dysfunction during physiological and pathological aging in humans.
Conflict of interest statement
Figures


Comment in
-
Neurogenesis altered in multiple neurodegenerative diseases.Nat Rev Neurol. 2021 Dec;17(12):726. doi: 10.1038/s41582-021-00588-4. Nat Rev Neurol. 2021. PMID: 34732827 No abstract available.
-
Adult neurogenesis in neurological diseases.Science. 2021 Nov 26;374(6571):1049-1050. doi: 10.1126/science.abm7468. Epub 2021 Nov 25. Science. 2021. PMID: 34822282
-
Adult neurogenesis is altered in neurodegenerative disease.Nat Neurosci. 2021 Dec;24(12):1640. doi: 10.1038/s41593-021-00978-3. Nat Neurosci. 2021. PMID: 34848879 No abstract available.
-
Comment on "Impact of neurodegenerative diseases on human adult hippocampal neurogenesis".Science. 2022 Apr 15;376(6590):eabn8861. doi: 10.1126/science.abn8861. Epub 2022 Apr 15. Science. 2022. PMID: 35420933 Free PMC article.
-
Comment on "Impact of neurodegenerative diseases on human adult hippocampal neurogenesis".Science. 2022 Apr 15;376(6590):eabn7083. doi: 10.1126/science.abn7083. Epub 2022 Apr 15. Science. 2022. PMID: 35420939 No abstract available.
References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

