The Expanding Role of Pyridine and Dihydropyridine Scaffolds in Drug Design
- PMID: 34675489
- PMCID: PMC8520849
- DOI: 10.2147/DDDT.S329547
The Expanding Role of Pyridine and Dihydropyridine Scaffolds in Drug Design
Abstract
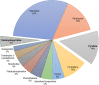
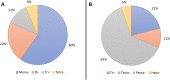
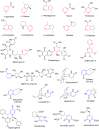
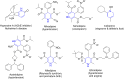
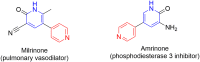
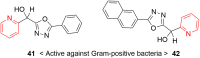
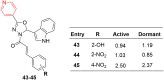
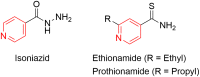
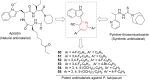
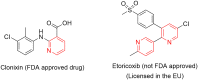
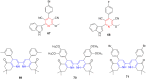
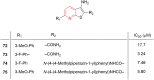
Pyridine-based ring systems are one of the most extensively used heterocycles in the field of drug design, primarily due to their profound effect on pharmacological activity, which has led to the discovery of numerous broad-spectrum therapeutic agents. In the US FDA database, there are 95 approved pharmaceuticals that stem from pyridine or dihydropyridine, including isoniazid and ethionamide (tuberculosis), delavirdine (HIV/AIDS), abiraterone acetate (prostate cancer), tacrine (Alzheimer's), ciclopirox (ringworm and athlete's foot), crizotinib (cancer), nifedipine (Raynaud's syndrome and premature birth), piroxicam (NSAID for arthritis), nilvadipine (hypertension), roflumilast (COPD), pyridostigmine (myasthenia gravis), and many more. Their remarkable therapeutic applications have encouraged researchers to prepare a larger number of biologically active compounds decorated with pyridine or dihydropyridine, expandeing the scope of finding a cure for other ailments. It is thus anticipated that myriad new pharmaceuticals containing the two heterocycles will be available in the forthcoming decade. This review examines the prospects of highly potent bioactive molecules to emphasize the advantages of using pyridine and dihydropyridine in drug design. We cover the most recent developments from 2010 to date, highlighting the ever-expanding role of both scaffolds in the field of medicinal chemistry and drug development.
Keywords: bioactive compounds; current trend; nitrogen heterocycles; pharmaceuticals; substituent effect.
© 2021 Ling et al.
Conflict of interest statement
The authors declare no conflicts of interest in this work.
Figures















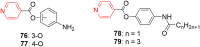
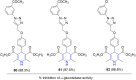
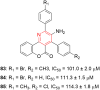
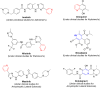
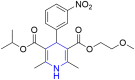




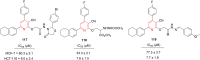
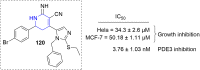
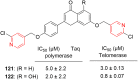
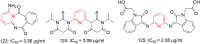
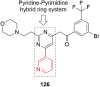
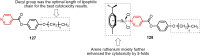


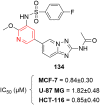
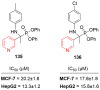
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

