A Marine λ-Oligocarrageenan Inhibits Migratory and Invasive Ability of MDA-MB-231 Human Breast Cancer Cells through Actions on Heparanase Metabolism and MMP-14/MMP-2 Axis
- PMID: 34677445
- PMCID: PMC8539239
- DOI: 10.3390/md19100546
A Marine λ-Oligocarrageenan Inhibits Migratory and Invasive Ability of MDA-MB-231 Human Breast Cancer Cells through Actions on Heparanase Metabolism and MMP-14/MMP-2 Axis
Abstract
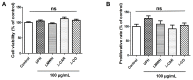
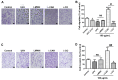
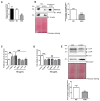
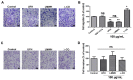
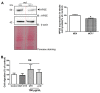
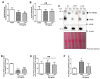
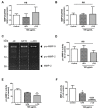
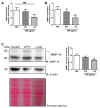
Sugar-based molecules such as heparins or natural heparan sulfate polysaccharides have been developed and widely studied for controlling heparanase (HPSE) enzymatic activity, a key player in extracellular matrix remodelling during cancer pathogenesis. However, non-enzymatic functions of HPSE have also been described in tumour mechanisms. Given their versatile properties, we hypothesized that sugar-based inhibitors may interfere with enzymatic but also non-enzymatic HPSE activities. In this work, we assessed the effects of an original marine λ-carrageenan derived oligosaccharide (λ-CO) we previously described, along with those of its native counterpart and heparins, on cell viability, proliferation, migration, and invasion of MDA-MB-231 breast cancer cells but also of sh-MDA-MB-231 cells, in which the expression of HPSE was selectively downregulated. We observed no cytotoxic and no anti-proliferative effects of our compounds but surprisingly λ-CO was the most efficient to reduce cell migration and invasion compared with heparins, and in a HPSE-dependent manner. We provided evidence that λ-CO tightly controlled a HPSE/MMP-14/MMP-2 axis, leading to reduced MMP-2 activity. Altogether, this study highlights λ-CO as a potent HPSE "modulator" capable of reducing not only the enzymatic activity of HPSE but also the functions controlled by the HPSE levels.
Keywords: MDA-MB-231; MMP-14; breast cancer; heparanase; heparin; metalloproteinase; oligosaccharide; polysaccharide; shRNA; λ-carrageenan.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
A Screening Approach to Assess the Impact of Various Commercial Sources of Crude Marine λ-Carrageenan on the Production of Oligosaccharides with Anti-heparanase and Anti-migratory Activities.Mar Drugs. 2023 May 11;21(5):295. doi: 10.3390/md21050295. Mar Drugs. 2023. PMID: 37233489 Free PMC article.
-
λ-Carrageenan Oligosaccharides of Distinct Anti-Heparanase and Anticoagulant Activities Inhibit MDA-MB-231 Breast Cancer Cell Migration.Mar Drugs. 2019 Feb 27;17(3):140. doi: 10.3390/md17030140. Mar Drugs. 2019. PMID: 30818840 Free PMC article.
-
Newly generated heparanase knock-out mice unravel co-regulation of heparanase and matrix metalloproteinases.PLoS One. 2009;4(4):e5181. doi: 10.1371/journal.pone.0005181. Epub 2009 Apr 10. PLoS One. 2009. PMID: 19360105 Free PMC article.
-
Antitumor activity and structure-activity relationship of heparanase inhibitors: Recent advances.Eur J Med Chem. 2020 May 1;193:112221. doi: 10.1016/j.ejmech.2020.112221. Epub 2020 Mar 14. Eur J Med Chem. 2020. PMID: 32222663 Review.
-
Recent data concerning heparanase: focus on fibrosis, inflammation and cancer.Biomol Concepts. 2015 Dec;6(5-6):415-21. doi: 10.1515/bmc-2015-0021. Biomol Concepts. 2015. PMID: 26552066 Review.
Cited by
-
Polysaccharides from marine biological resources and their anticancer activity on breast cancer.RSC Med Chem. 2023 Apr 13;14(6):1049-1059. doi: 10.1039/d3md00035d. eCollection 2023 Jun 22. RSC Med Chem. 2023. PMID: 37360387 Free PMC article. Review.
-
Natural-Product-Derived Adjunctive Treatments to Conventional Therapy and Their Immunoregulatory Activities in Triple-Negative Breast Cancer.Molecules. 2023 Aug 1;28(15):5804. doi: 10.3390/molecules28155804. Molecules. 2023. PMID: 37570775 Free PMC article. Review.
-
A Screening Approach to Assess the Impact of Various Commercial Sources of Crude Marine λ-Carrageenan on the Production of Oligosaccharides with Anti-heparanase and Anti-migratory Activities.Mar Drugs. 2023 May 11;21(5):295. doi: 10.3390/md21050295. Mar Drugs. 2023. PMID: 37233489 Free PMC article.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

