Linking the Defective Structure of Boron-Doped Carbon Nano-Onions with Their Catalytic Properties: Experimental and Theoretical Studies
- PMID: 34677930
- PMCID: PMC8569677
- DOI: 10.1021/acsami.1c12126
Linking the Defective Structure of Boron-Doped Carbon Nano-Onions with Their Catalytic Properties: Experimental and Theoretical Studies
Abstract
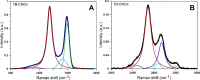
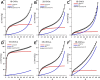
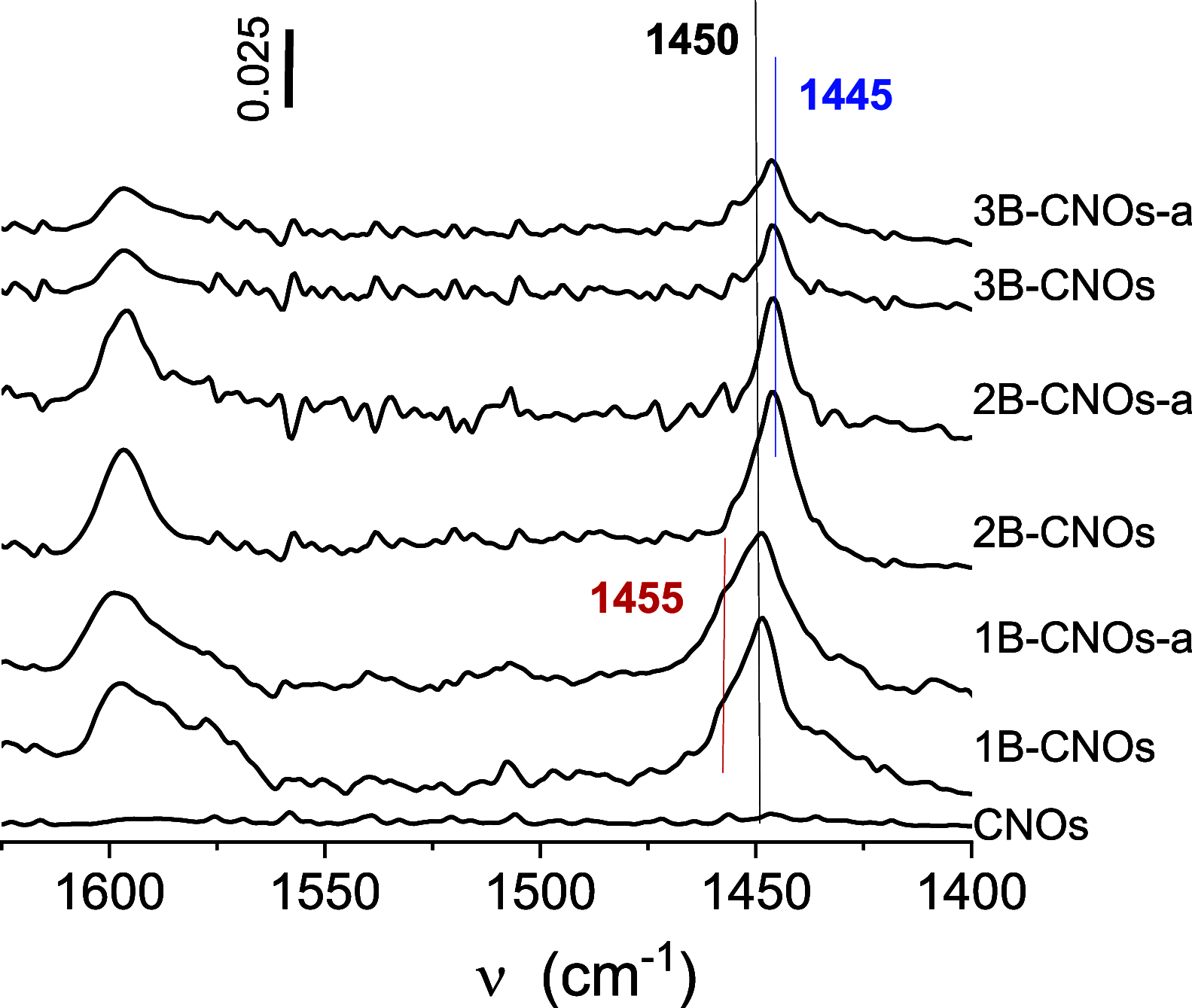
Defects are widely present in nanomaterials, and they are recognized as the active sites that tune surface properties in the local region for catalysis. Recently, the theory linking defect structures and catalytic properties of nanocatalysts has been most commonly described. In this study, we prepared boron-doped carbon nano-onions (B-CNOs) by applying an annealing treatment of ultradispersed nanodiamond particles and amorphous boron. These experimental conditions guarantee doping of CNOs with boron atoms in the entire carbon nanostructure, thereby ensuring structural homogeneity. In our research, we discuss the correlations between defective structures of B-CNOs with their catalytic properties toward SO2 and tert-butanol dehydration. We show that there is a close relationship between the catalytic properties of the B-CNOs and the experimental conditions for their formation. It is not only the mass of the substrates used for the formation of B-CNOs that is crucial, that is, the mass ratio of NDs to amorphous B, but also the process, including temperature and gas atmosphere. As it was expected, all B-CNOs demonstrated significant catalytic activity in HSO3- oxidation. However, the subsequent annealing in an air atmosphere diminished their catalytic activity. Unfortunately, no direct relationship between the catalytic activity and the presence of heteroatoms on the B-CNO surface was observed. There was a linear dependence between catalytic activity and Raman reactivity factors for each of the B-CNO materials. In contrast to SO2 oxidation, the B-CNO-a samples showed higher catalytic activity in tert-butanol dehydration due to the presence of Brønsted and Lewis acid sites. The occurence of three types of boron-Lewis sites differing in electron donor properties was confirmed using quantitative infrared spectroscopic measurements of pyridine adsorption.
Keywords: boron; carbon nano-onion; carbon nanostructure; catalysis; defect; doping.
Conflict of interest statement
The authors declare no competing financial interest.
Figures






Similar articles
-
Boron-Doped Polygonal Carbon Nano-Onions: Synthesis and Applications in Electrochemical Energy Storage.Chemistry. 2017 May 23;23(29):7132-7141. doi: 10.1002/chem.201700914. Epub 2017 Apr 26. Chemistry. 2017. PMID: 28339126
-
Platinum electrodeposition on unsupported carbon nano-onions.Langmuir. 2012 Dec 11;28(49):17202-10. doi: 10.1021/la3031396. Epub 2012 Nov 26. Langmuir. 2012. PMID: 23145813 Free PMC article.
-
In Vitro and In Vivo Biocompatibility of Boron/Nitrogen Co-Doped Carbon Nano-Onions.Nanomaterials (Basel). 2021 Nov 10;11(11):3017. doi: 10.3390/nano11113017. Nanomaterials (Basel). 2021. PMID: 34835781 Free PMC article.
-
Application of carbon nano onions in the biomedical field: recent advances and challenges.Biomater Sci. 2021 Feb 9;9(3):626-644. doi: 10.1039/d0bm01476a. Biomater Sci. 2021. PMID: 33241797 Review.
-
Carbon nano-onions (multi-layer fullerenes): chemistry and applications.Beilstein J Nanotechnol. 2014 Nov 4;5:1980-98. doi: 10.3762/bjnano.5.207. eCollection 2014. Beilstein J Nanotechnol. 2014. PMID: 25383308 Free PMC article. Review.
Cited by
-
Microwave-Assisted Synthesis as a Promising Tool for the Preparation of Materials Containing Defective Carbon Nanostructures: Implications on Properties and Applications.Materials (Basel). 2023 Oct 4;16(19):6549. doi: 10.3390/ma16196549. Materials (Basel). 2023. PMID: 37834689 Free PMC article. Review.
References
-
- Kotakoski J.; Krasheninnikov A. V.; Kaiser U.; Meyer J. C.. From Point Defects in Graphene to Two-Dimensional Amorphous Carbon. Phys. Rev. Lett. 2011, 106(). https://doi.org/10.1103/PhysRevLett.106.105505.10.1103/PhysRevLett.106.1... - DOI - PubMed
-
- Jia Y.; Jiang K.; Wang H.; Yao X. The Role of Defect Sites in Nanomaterials for Electrocatalytic Energy Conversion. Chem 2019, 5, 1371–1397. 10.1016/j.chempr.2019.02.008. - DOI
-
- Yang H. B.; Miao J.; Hung S.-F.; Chen J.; Tao H. B.; Wang X.; Zhang L.; Chen R.; Gao J.; Chen H. M.; Dai L.; Liu B. Identification of Catalytic Sites for Oxygen Reduction and Oxygen Evolution in N-Doped Graphene Materials: Development of Highly Efficient Metal-Free Bifunctional Electrocatalyst. Sci. Adv. 2016, 2, e150112210.1126/sciadv.1501122. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

